Defining the neural fulcrum for chronic vagus nerve stimulation: implications for integrated cardiac control
- PMID: 28862330
- PMCID: PMC5685838
- DOI: 10.1113/JP274678
Defining the neural fulcrum for chronic vagus nerve stimulation: implications for integrated cardiac control
Abstract
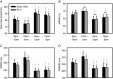
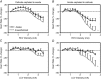
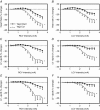
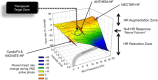
Key points: The evoked cardiac response to bipolar cervical vagus nerve stimulation (VNS) reflects a dynamic interaction between afferent mediated decreases in central parasympathetic drive and suppressive effects evoked by direct stimulation of parasympathetic efferent axons to the heart. The neural fulcrum is defined as the operating point, based on frequency-amplitude-pulse width, where a null heart rate response is reproducibly evoked during the on-phase of VNS. Cardiac control, based on the principal of the neural fulcrum, can be elicited from either vagus. Beta-receptor blockade does not alter the tachycardia phase to low intensity VNS, but can increase the bradycardia to higher intensity VNS. While muscarinic cholinergic blockade prevented the VNS-induced bradycardia, clinically relevant doses of ACE inhibitors, beta-blockade and the funny channel blocker ivabradine did not alter the VNS chronotropic response. While there are qualitative differences in VNS heart control between awake and anaesthetized states, the physiological expression of the neural fulcrum is maintained.
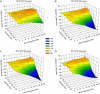
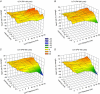
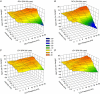
Abstract: Vagus nerve stimulation (VNS) is an emerging therapy for treatment of chronic heart failure and remains a standard of therapy in patients with treatment-resistant epilepsy. The objective of this work was to characterize heart rate (HR) responses (HRRs) during the active phase of chronic VNS over a wide range of stimulation parameters in order to define optimal protocols for bidirectional bioelectronic control of the heart. In normal canines, bipolar electrodes were chronically implanted on the cervical vagosympathetic trunk bilaterally with anode cephalad to cathode (n = 8, 'cardiac' configuration) or with electrode positions reversed (n = 8, 'epilepsy' configuration). In awake state, HRRs were determined for each combination of pulse frequency (2-20 Hz), intensity (0-3.5 mA) and pulse widths (130-750 μs) over 14 months. At low intensities and higher frequency VNS, HR increased during the VNS active phase owing to afferent modulation of parasympathetic central drive. When functional effects of afferent and efferent fibre activation were balanced, a null HRR was evoked (defined as 'neural fulcrum') during which HRR ≈ 0. As intensity increased further, HR was reduced during the active phase of VNS. While qualitatively similar, VNS delivered in the epilepsy configuration resulted in more pronounced HR acceleration and reduced HR deceleration during VNS. At termination, under anaesthesia, transection of the vagi rostral to the stimulation site eliminated the augmenting response to VNS and enhanced the parasympathetic efferent-mediated suppressing effect on electrical and mechanical function of the heart. In conclusion, VNS activates central then peripheral aspects of the cardiac nervous system. VNS control over cardiac function is maintained during chronic therapy.
Keywords: cardiac afferent; neural fulcrum; neurocardiology; parasympathetic; vagus nerve stimulation.
© 2017 The Authors. The Journal of Physiology © 2017 The Physiological Society.
Figures



Comment in
-
The comings and goings of the vagus and the need to know your neural fulcrum.J Physiol. 2017 Nov 15;595(22):6809-6810. doi: 10.1113/JP275204. Epub 2017 Oct 19. J Physiol. 2017. PMID: 29052217 Free PMC article. No abstract available.
References
-
- Anholt TA, Ayal S & Goldberg JA (2011). Recruitment and blocking properties of the CardioFit stimulation lead. J Neural Eng 8, 034004. - PubMed
-
- Ardell JL & Armour JA (2016). Neurocardiology: structure‐based function. Compr Physiol 6, 1635–1653. - PubMed
-
- Arle JE, Carlson KW & Mei L (2016). Investigation of mechanisms of vagus nerve stimulation for seizure using finite element modeling. Epilepsy Res 126, 109–118. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

