Vinexin family (SORBS) proteins play different roles in stiffness-sensing and contractile force generation
- PMID: 28864765
- PMCID: PMC5665443
- DOI: 10.1242/jcs.200691
Vinexin family (SORBS) proteins play different roles in stiffness-sensing and contractile force generation
Abstract
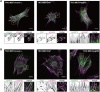
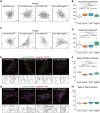
Vinexin, c-Cbl associated protein (CAP) and Arg-binding protein 2 (ArgBP2) constitute an adaptor protein family called the vinexin (SORBS) family that is targeted to focal adhesions (FAs). Although numerous studies have focused on each of the SORBS proteins and partially elucidated their involvement in mechanotransduction, a comparative analysis of their function has not been well addressed. Here, we established mouse embryonic fibroblasts that individually expressed SORBS proteins and analysed their functions in an identical cell context. Both vinexin-α and CAP co-localized with vinculin at FAs and promoted the appearance of vinculin-rich FAs, whereas ArgBP2 co-localized with α-actinin at the proximal end of FAs and punctate structures on actin stress fibers (SFs), and induced paxillin-rich FAs. Furthermore, both vinexin-α and CAP contributed to extracellular matrix stiffness-dependent vinculin behaviors, while ArgBP2 stabilized α-actinin on SFs and enhanced intracellular contractile forces. These results demonstrate the differential roles of SORBS proteins in mechanotransduction.
Keywords: Actin cytoskeleton; ArgBP2; CAP; Focal adhesion; Mechanotransduction; Vinexin.
© 2017. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures






References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

