Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp
- PMID: 28867785
- PMCID: PMC5618522
- DOI: 10.3390/ijms18091873
Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp
Abstract
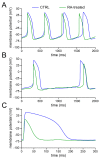
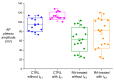
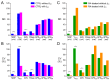
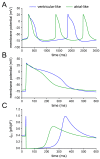
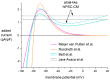
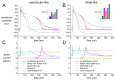
Human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) hold great promise for studying inherited cardiac arrhythmias and developing drug therapies to treat such arrhythmias. Unfortunately, until now, action potential (AP) measurements in hiPSC-CMs have been hampered by the virtual absence of the inward rectifier potassium current (IK1) in hiPSC-CMs, resulting in spontaneous activity and altered function of various depolarising and repolarising membrane currents. We assessed whether AP measurements in "ventricular-like" and "atrial-like" hiPSC-CMs could be improved through a simple, highly reproducible dynamic clamp approach to provide these cells with a substantial IK1 (computed in real time according to the actual membrane potential and injected through the patch-clamp pipette). APs were measured at 1 Hz using perforated patch-clamp methodology, both in control cells and in cells treated with all-trans retinoic acid (RA) during the differentiation process to increase the number of cells with atrial-like APs. RA-treated hiPSC-CMs displayed shorter APs than control hiPSC-CMs and this phenotype became more prominent upon addition of synthetic IK1 through dynamic clamp. Furthermore, the variability of several AP parameters decreased upon IK1 injection. Computer simulations with models of ventricular-like and atrial-like hiPSC-CMs demonstrated the importance of selecting an appropriate synthetic IK1. In conclusion, the dynamic clamp-based approach of IK1 injection has broad applicability for detailed AP measurements in hiPSC-CMs.
Keywords: cardiomyocytes; computer simulations; differentiation; electro-physiology; induced pluripotent stem cells; inward rectifier potassium current; perforated patch-clamp; retinoic acid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Meijer van Putten R.M.E., Mengarelli I., Guan K., Zegers J.G., van Ginneken A.C.G., Verkerk A.O., Wilders R. Ion channelopathies in human induced pluripotent stem cell derived cardiomyocytes: A dynamic clamp study with virtual IK1. Front. Physiol. 2015;6:7. doi: 10.3389/fphys.2015.00007. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

