Cell migration, intercalation and growth regulate mammalian cochlear extension
- PMID: 28870992
- PMCID: PMC5675446
- DOI: 10.1242/dev.151761
Cell migration, intercalation and growth regulate mammalian cochlear extension
Abstract
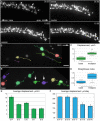
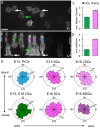
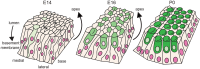
Developmental remodeling of the sensory epithelium of the cochlea is required for the formation of an elongated, tonotopically organized auditory organ, but the cellular processes that mediate these events are largely unknown. We used both morphological assessments of cellular rearrangements and time-lapse imaging to visualize cochlear remodeling in mouse. Analysis of cell redistribution showed that the cochlea extends through a combination of radial intercalation and cell growth. Live imaging demonstrated that concomitant cellular intercalation results in a brief period of epithelial convergence, although subsequent changes in cell size lead to medial-lateral spreading. Supporting cells, which retain contact with the basement membrane, exhibit biased protrusive activity and directed movement along the axis of extension. By contrast, hair cells lose contact with the basement membrane, but contribute to continued outgrowth through increased cell size. Regulation of cellular protrusions, movement and intercalation within the cochlea all require myosin II. These results establish, for the first time, many of the cellular processes that drive the distribution of sensory cells along the tonotopic axis of the cochlea.
Keywords: Cochlear development; Convergent extension; Inner ear; Live imaging; Mouse; Radial intercalation.
© 2017. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

