Autophagy in diabetic kidney disease: regulation, pathological role and therapeutic potential
- PMID: 28871310
- PMCID: PMC5771948
- DOI: 10.1007/s00018-017-2639-1
Autophagy in diabetic kidney disease: regulation, pathological role and therapeutic potential
Abstract
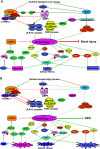
Diabetic kidney disease, a leading cause of end-stage renal disease, has become a serious public health problem worldwide and lacks effective therapies. Autophagy is a highly conserved lysosomal degradation pathway that removes protein aggregates and damaged organelles to maintain cellular homeostasis. As important stress-responsive machinery, autophagy is involved in the pathogenesis of various diseases. Emerging evidence has suggested that dysregulated autophagy may contribute to both glomerular and tubulointerstitial pathologies in kidneys under diabetic conditions. This review summarizes the recent findings regarding the role of autophagy in the pathogenesis of diabetic kidney disease and highlights the regulation of autophagy by the nutrient-sensing pathways and intracellular stress signaling in this disease. The advances in our understanding of autophagy in diabetic kidney disease will facilitate the discovery of a new therapeutic target for the prevention and treatment of this life-threatening diabetes complication.
Keywords: AMPK; ER stress; Hypoxia; Reactive oxygen species; Sirt1; mTOR.
Figures
References
-
- USRDS USRDS: the United States Renal Data System. Am J Kidney Dis. 2003;42(6 Suppl 5):1–230. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

