Intersectin-1s deficiency in pulmonary pathogenesis
- PMID: 28874189
- PMCID: PMC5585975
- DOI: 10.1186/s12931-017-0652-4
Intersectin-1s deficiency in pulmonary pathogenesis
Abstract
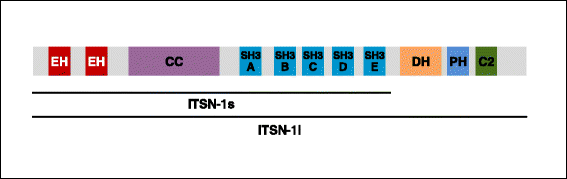
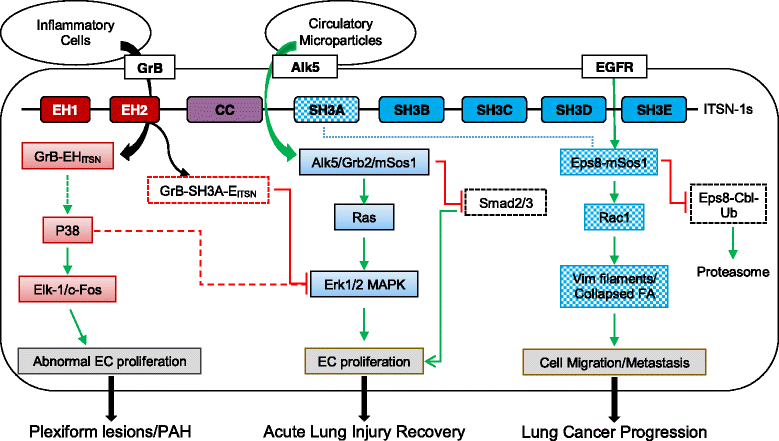
Intersectin-1s (ITSN-1s), a multidomain adaptor protein, plays a vital role in endocytosis, cytoskeleton rearrangement and cell signaling. Recent studies have demonstrated that deficiency of ITSN-1s is a crucial early event in pulmonary pathogenesis. In lung cancer, ITSN-1s deficiency impairs Eps8 ubiquitination and favors Eps8-mSos1 interaction which activates Rac1 leading to enhanced lung cancer cell proliferation, migration and metastasis. Restoring ITSN-1s deficiency in lung cancer cells facilitates cytoskeleton changes favoring mesenchymal to epithelial transformation and impairs lung cancer progression. ITSN-1s deficiency in acute lung injury leads to impaired endocytosis which leads to ubiquitination and degradation of growth factor receptors such as Alk5. This deficiency is counterbalanced by microparticles which, via paracrine effects, transfer Alk5/TGFβRII complex to non-apoptotic cells. In the presence of ITSN-1s deficiency, Alk5-restored cells signal via Erk1/2 MAPK pathway leading to restoration and repair of lung architecture. In inflammatory conditions such as pulmonary artery hypertension, ITSN-1s full length protein is cleaved by granzyme B into EHITSN and SH3A-EITSN fragments. The EHITSN fragment leads to pulmonary cell proliferation via activation of p38 MAPK and Elk-1/c-Fos signaling. In vivo, ITSN-1s deficient mice transduced with EHITSN plasmid develop pulmonary vascular obliteration and plexiform lesions consistent with pathological findings seen in severe pulmonary arterial hypertension. These novel findings have significantly contributed to understanding the mechanisms and pathogenesis involved in pulmonary pathology. As demonstrated in these studies, genetically modified ITSN-1s expression mouse models will be a valuable tool to further advance our understanding of pulmonary pathology and lead to novel targets for treating these conditions.
Keywords: Acute lung injury; Eps8; Intersectin-1s; Lung cancer; MAPK; Pulmonary arterial hypertension; Rac1; mSos1.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interest.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

