Mitochondrial permeability transition pore: sensitivity to opening and mechanistic dependence on substrate availability
- PMID: 28874733
- PMCID: PMC5585167
- DOI: 10.1038/s41598-017-10673-8
Mitochondrial permeability transition pore: sensitivity to opening and mechanistic dependence on substrate availability
Abstract
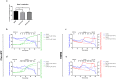
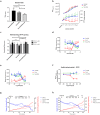
Mitochondrial Ca2+ uptake has a key role in cellular Ca2+ homeostasis. Excessive matrix Ca2+ concentrations, especially when coincident with oxidative stress, precipitate opening of an inner mitochondrial membrane, high-conductance channel: the mitochondrial permeability transition pore (mPTP). mPTP opening has been implicated as a final cell death pathway in numerous diseases and therefore understanding conditions dictating mPTP opening is crucial for developing targeted therapies. Here, we have investigated the impact of mitochondrial metabolic state on the probability and consequences of mPTP opening. Isolated mitochondria were energised using NADH- or FADH2-linked substrates. The functional consequences of Ca2+-induced mPTP opening were assessed by Ca2+ retention capacity, using fluorescence-based analysis, and simultaneous measurements of mitochondrial Ca2+ handling, membrane potential, respiratory rate and production of reactive oxygen species (ROS). Succinate-induced, membrane potential-dependent reverse electron transfer sensitised mitochondria to mPTP opening. mPTP-induced depolarisation under succinate subsequently inhibited reverse electron transfer. Complex I-driven respiration was reduced after mPTP opening but sustained in the presence of complex II-linked substrates, consistent with inhibition of complex I-supported respiration by leakage of matrix NADH. Additionally, ROS generated at complex III did not sensitise mitochondria to mPTP opening. Thus, cellular metabolic fluxes and metabolic environment dictate mitochondrial functional response to Ca2+ overload.
Conflict of interest statement
T.B., M.R., S.L., B.P. and J.S. are employees of Eisai.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

