Bioorthogonal Cycloadditions: Computational Analysis with the Distortion/Interaction Model and Predictions of Reactivities
- PMID: 28876890
- PMCID: PMC5675536
- DOI: 10.1021/acs.accounts.7b00265
Bioorthogonal Cycloadditions: Computational Analysis with the Distortion/Interaction Model and Predictions of Reactivities
Abstract
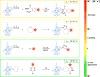
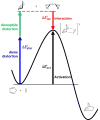
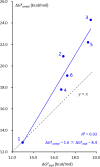
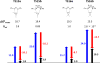
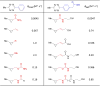
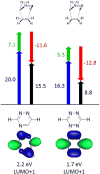
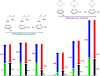
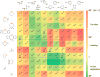
Bioorthogonal chemistry has had a major impact on the study of biological processes in vivo. Biomolecules of interest can be tracked by using probes and reporters that do not react with cellular components and do not interfere with metabolic processes in living cells. Much time and effort has been devoted to the screening of potential bioorthogonal reagents experimentally. This Account describes how our groups have performed computational screening of reactivity and mutual orthogonality. Our collaborations with experimentalists have led to the development of new and useful reactions. Dozens of bioorthogonal cycloadditions have been reported in the literature in the past few years, but as interest in tracking multiple targets arises, our computational screening has gained importance for the discovery of new mutually orthogonal bioorthogonal cycloaddition pairs. The reactivities of strained alkenes and alkynes with common 1,3-dipoles such as azides, along with mesoionic sydnones and other novel 1,3-dipoles, have been explored. Studies of "inverse-electron-demand" dienes such as triazines and tetrazines that have been used in bioorthogonal Diels-Alder cycloadditions are described. The color graphics we have developed give a snapshot of whether reactions are fast enough for cellular applications (green), adequately reactive for labeling (yellow), or only useful for synthesis or do not occur at all (red). The colors of each box give an instant view of rates, while bar graphs provide an analysis of the factors that control reactivity. This analysis uses the distortion/interaction or activation strain model of cycloaddition reactivity developed independently by our group and that of F. Matthias Bickelhaupt in The Netherlands. The model analyzes activation barriers in terms of the energy required to distort the reactants to the transition state geometry. This energy, called the distortion energy or activation strain, constitutes the major component of the activation energy. The strong bonding interaction between the termini of the two reactants, which we call the interaction energy, overcomes the distortion energy and leads to the new bonds in the products. This Account describes how we have analyzed and predicted bioorthogonal cycloaddition reactivity using the distortion/interaction model and how our experimental collaborators have employed these insights to create new bioorthogonal cycloadditions. The graphics we use document and predict which combinations of cycloadditions will be useful in bioorthogonal chemistry and which pairs of reactions are mutually orthogonal. For example, the fast reaction of 5-phenyl-1,2,4-triazine and a thiacycloheptyne will not interfere with the other fast reaction of 3,6-diphenyl-1,2,4,5-tetrazine and a cyclopropene. No cross reactions will occur, as these are very slow reactions.
Figures
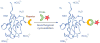



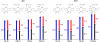

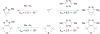
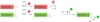
Similar articles
-
Constructing New Bioorthogonal Reagents and Reactions.Acc Chem Res. 2018 May 15;51(5):1073-1081. doi: 10.1021/acs.accounts.7b00606. Epub 2018 May 4. Acc Chem Res. 2018. PMID: 29727171 Free PMC article.
-
Electrophilic Azides for Materials Synthesis and Chemical Biology.Acc Chem Res. 2020 Apr 21;53(4):937-948. doi: 10.1021/acs.accounts.0c00046. Epub 2020 Mar 24. Acc Chem Res. 2020. PMID: 32207916 Review.
-
Synthesis and reactivity comparisons of 1-methyl-3-substituted cyclopropene mini-tags for tetrazine bioorthogonal reactions.Chemistry. 2014 Mar 17;20(12):3365-75. doi: 10.1002/chem.201304225. Epub 2014 Feb 24. Chemistry. 2014. PMID: 24615990 Free PMC article.
-
Advances in Tetrazine Bioorthogonal Chemistry Driven by the Synthesis of Novel Tetrazines and Dienophiles.Acc Chem Res. 2018 May 15;51(5):1249-1259. doi: 10.1021/acs.accounts.8b00062. Epub 2018 Apr 11. Acc Chem Res. 2018. PMID: 29638113 Free PMC article.
-
Computational Organic Chemistry: The Frontier for Understanding and Designing Bioorthogonal Cycloadditions.Top Curr Chem (Cham). 2024 May 10;382(2):17. doi: 10.1007/s41061-024-00461-0. Top Curr Chem (Cham). 2024. PMID: 38727989 Free PMC article. Review.
Cited by
-
Discovery of new tetrazines for bioorthogonal reactions with strained alkenes via computational chemistry.RSC Adv. 2024 Jan 31;14(7):4345-4351. doi: 10.1039/d3ra08712c. eCollection 2024 Jan 31. RSC Adv. 2024. PMID: 38304564 Free PMC article.
-
Click-Ready Perfluorocarbon Nanoemulsion for 19F MRI and Multimodal Cellular Detection.ACS Nanosci Au. 2022 Apr 20;2(2):102-110. doi: 10.1021/acsnanoscienceau.1c00016. Epub 2021 Nov 11. ACS Nanosci Au. 2022. PMID: 35481225 Free PMC article.
-
Labeling a TCO-functionalized single domain antibody fragment with 18F via inverse electron demand Diels Alder cycloaddition using a fluoronicotinyl moiety-bearing tetrazine derivative.Bioorg Med Chem. 2020 Sep 1;28(17):115634. doi: 10.1016/j.bmc.2020.115634. Epub 2020 Jul 9. Bioorg Med Chem. 2020. PMID: 32773089 Free PMC article.
-
Photo-controllable bioorthogonal chemistry for spatiotemporal control of bio-targets in living systems.Chem Sci. 2020 Mar 10;11(13):3390-3396. doi: 10.1039/c9sc06540g. Chem Sci. 2020. PMID: 34109018 Free PMC article. Review.
-
Mutually Orthogonal Bioorthogonal Reactions: Selective Chemistries for Labeling Multiple Biomolecules Simultaneously.Top Curr Chem (Cham). 2024 Jul 6;382(3):24. doi: 10.1007/s41061-024-00467-8. Top Curr Chem (Cham). 2024. PMID: 38971884 Free PMC article. Review.
References
-
- Kobayashi S, Jorgensen KA. Cycloaddition Reactions in Organic Synthesis. Wiley-VCH; Weinheim, Germany: 2001.
- Nishiwaki N. Methods and Applications of Cycloaddition Reactions in Organic Synthesis. Wiley-VCH; Weinheim, Germany: 2014.
- Nicolaou KC, Vourloumis D, Winssinger N, Baran PS. The Art and Science of Total Synthesis. Angew. Chem. Int. Ed. 2000;39:44–122. - PubMed
-
- Huisgen R. 1,3-Dipolar Cycloadditions. Past and Future. Angew. Chem. Int. Ed. Engl. 1963;2:565–632.
-
- Diels O, Alder K. Synthesen in der Hydroaromatischen Reihe. Justus Liebigs Annalen der Chemie. 1928;460:98.
-
- Houk KN, Sims J, Duke RE, Jr, Strozier RW, George JK. Frontier Molecular Orbitals of 1,3-Dipoles and Dipolarphiles. J. Am. Chem. Soc. 1973;95:7287–7300.
- Houk KN, Sims J, Watts CR, Luskus LJ. The Origin of Reactivity, Regioselectivity, and Periselectivity in 1,3-Dipolar Cycloadditions. J. Am. Chem. Soc. 1973;95:7301–7315.
- Houk KN. Frontier Molecular Orbital Theory of Cycloaddition Reactions. Acc. Chem. Res. 1975;8:361–369.
- Houk KN. Organic Chemistry. Springer; Berlin: 1979. Theoretical and Experimental Insights into Cycloaddition Reactions; pp. 1–40.
- Houk KN, Yamaguchi K. Theory of 1,3-Dipolar Cycloadditions. In: Padwa A, editor. 1,3-Dipolar Cycloaddition Chemistry. Vol. 2. Wiley; New York: 1984. pp. 407–450.
- Ess DH, Jones GO, Houk KN. Conceptual, Qualitative, and Quantitative Theories of 1,3-Dipolar and Diels–Alder Cycloadditions Used in Synthesis. Adv. Synth. Catal. 2006;348:2337–2361.
-
- Ess DH, Houk KN. Distortion/Interaction Energy Control of 1,3-Dipolar Cycloaddition Reactivity. J. Am. Chem. Soc. 2007;129:10646–10647. - PubMed
- Ess DH, Houk KN. Theory of 1,3-Dipolar Cycloadditions: Distortion/Interaction and Frontier Molecular Orbital Models. J. Am. Chem. Soc. 2008;130:10187–10198. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

