Impairments in age-dependent ubiquitin proteostasis and structural integrity of selective neurons by uncoupling Ran GTPase from the Ran-binding domain 3 of Ranbp2 and identification of novel mitochondrial isoforms of ubiquitin-conjugating enzyme E2I (ubc9) and Ranbp2
- PMID: 28877029
- PMCID: PMC6380291
- DOI: 10.1080/21541248.2017.1356432
Impairments in age-dependent ubiquitin proteostasis and structural integrity of selective neurons by uncoupling Ran GTPase from the Ran-binding domain 3 of Ranbp2 and identification of novel mitochondrial isoforms of ubiquitin-conjugating enzyme E2I (ubc9) and Ranbp2
Abstract
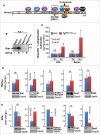
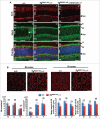
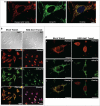
The Ran-binding protein 2 (Ranbp2/Nup358) is a cytoplasmic and peripheral nucleoporin comprised of 4 Ran-GTP-binding domains (RBDs) that are interspersed among diverse structural domains with multifunctional activities. Our prior studies found that the RBD2 and RBD3 of Ranbp2 control mitochondrial motility independently of Ran-GTP-binding in cultured cells, whereas loss of Ran-GTP-binding to RBD2 and RBD3 are essential to support cone photoreceptor development and the survival of mature retinal pigment epithelium (RPE) in mice. Here, we uncover that loss of Ran-GTP-binding to RBD3 alone promotes the robust age-dependent increase of ubiquitylated substrates and S1 subunit (Pmsd1) of the 19S cap of the proteasome in the retina and RPE and that such loss in RBD3 also compromises the structural integrity of the outer segment compartment of cone photoreceptors only and without affecting the viability of these neurons. We also found that the E2-ligase and partner of Ranbp2, ubc9, is localized prominently in the mitochondrial-rich ellipsoid compartment of photoreceptors, where Ranbp2 is also known to localize with and modulate the activity of mitochondrial proteins. However, the natures of Ranbp2 and ubc9 isoforms to the mitochondria are heretofore elusive. Subcellular fractionation, co-immunolocalization and immunoaffinity purification of Ranbp2 complexes show that novel isoforms of Ranbp2 and ubc9 with molecular masses distinct from the large Ranbp2 and unmodified ubc9 isoforms localize specifically to the mitochondrial fraction or associate with mitochondrial components, whereas unmodified and SUMOylated Ran GTPase are excluded from the mitochondrial fraction. Further, liposome-mediated intracellular delivery of an antibody against a domain shared by the mitochondrial and nuclear pore isoforms of Ranbp2 causes the profound fragmentation of mitochondria and their delocalization from Ranbp2 and without affecting Ranbp2 localization at the nuclear pores. Collectively, the data support that Ran GTPase-dependent and independent and moonlighting roles of Ranbp2 or domains thereof and ubc9 control selectively age-dependent, neural-type and mitochondrial functions.
Keywords: Ran GTPase; Ran-binding protein 2; mitochondria; nucleoporin; photoreceptor neuron; transgenic mice; ubiquitin proteostasis; ubiquitin-conjugating enzyme E2I (ubc9).
Figures



Similar articles
-
Selective impairment of a subset of Ran-GTP-binding domains of ran-binding protein 2 (Ranbp2) suffices to recapitulate the degeneration of the retinal pigment epithelium (RPE) triggered by Ranbp2 ablation.J Biol Chem. 2014 Oct 24;289(43):29767-89. doi: 10.1074/jbc.M114.586834. Epub 2014 Sep 3. J Biol Chem. 2014. PMID: 25187515 Free PMC article.
-
Structural basis for a nucleoporin exportin complex between RanBP2, SUMO1-RanGAP1, the E2 Ubc9, Crm1 and the Ran GTPase.Nat Commun. 2025 Jul 11;16(1):6403. doi: 10.1038/s41467-025-61694-1. Nat Commun. 2025. PMID: 40640138 Free PMC article.
-
Kinesin-1 and mitochondrial motility control by discrimination of structurally equivalent but distinct subdomains in Ran-GTP-binding domains of Ran-binding protein 2.Open Biol. 2013 Mar 27;3(3):120183. doi: 10.1098/rsob.120183. Open Biol. 2013. PMID: 23536549 Free PMC article.
-
Performing in vitro sumoylation reactions using recombinant enzymes.Methods Mol Biol. 2009;497:187-99. doi: 10.1007/978-1-59745-566-4_12. Methods Mol Biol. 2009. PMID: 19107418 Review.
-
RANBP2 evolution and human disease.FEBS Lett. 2023 Oct;597(20):2519-2533. doi: 10.1002/1873-3468.14749. Epub 2023 Oct 15. FEBS Lett. 2023. PMID: 37795679 Review.
Cited by
-
Role of oxidative stress in neurodegenerative disorders: a review of reactive oxygen species and prevention by antioxidants.Brain Commun. 2024 Jan 2;6(1):fcad356. doi: 10.1093/braincomms/fcad356. eCollection 2024. Brain Commun. 2024. PMID: 38214013 Free PMC article. Review.
-
Workshop on RanBP2/Nup358 and acute necrotizing encephalopathy.Nucleus. 2022 Dec;13(1):154-169. doi: 10.1080/19491034.2022.2069071. Nucleus. 2022. PMID: 35485383 Free PMC article.
-
Advances in the understanding of nuclear pore complexes in human diseases.J Cancer Res Clin Oncol. 2024 Jul 30;150(7):374. doi: 10.1007/s00432-024-05881-5. J Cancer Res Clin Oncol. 2024. PMID: 39080077 Free PMC article. Review.
-
Hyperosmolality in CHO cell culture: effects on the proteome.Appl Microbiol Biotechnol. 2022 Apr;106(7):2569-2586. doi: 10.1007/s00253-022-11861-x. Epub 2022 Mar 21. Appl Microbiol Biotechnol. 2022. PMID: 35312825 Free PMC article.
-
The coming-of-age of nucleocytoplasmic transport in motor neuron disease and neurodegeneration.Cell Mol Life Sci. 2019 Jun;76(12):2247-2273. doi: 10.1007/s00018-019-03029-0. Epub 2019 Feb 11. Cell Mol Life Sci. 2019. PMID: 30742233 Free PMC article. Review.
References
-
- Hamada M, Haeger A, Jeganathan KB, van Ree JH, Malureanu L, Walde S, Joseph J, Kehlenbach RH, van Deursen JM. Ran-dependent docking of importin-beta to RanBP2/Nup358 filaments is essential for protein import and cell viability. J Cell Biol. 2011;194(4):597-612. doi:10.1083/jcb.201102018. PMID:21859863 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
