Synthesis of Polymer-Mesoporous Silica Nanocomposites
- PMID: 28883321
- PMCID: PMC5445796
- DOI: 10.3390/ma3074066
Synthesis of Polymer-Mesoporous Silica Nanocomposites
Abstract
Polymer nanocomposites show unique properties combining the advantages of the inorganic nanofillers and the organic polymers. The mesoporous silica nanofillers have received much attention due to their ordered structure, high surface area and ease for functionalization of the nanopores. To accommodate macromolecules, the nanopores lead to unusually intimate interactions between the polymer and the inorganic phase, and some unusual properties can be observed, when compared with nonporous fillers. Whereas many review articles have been devoted to polymer/nonporous nanofiller nanocomposites, few review articles focus on polymer/mesoporous silica nanocomposites. This review summarizes the recent development in the methods for synthesizing polymer/mesoporous silica nanocomposites based on the papers published from 1998 to 2009, and some unique properties of these composites are also described.
Keywords: mesoporous silica; nanocomposites; polymer.
Figures

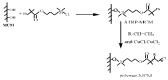
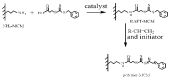

Similar articles
-
Thermally stable polymer composites with improved transparency by using colloidal mesoporous silica nanoparticles as inorganic fillers.Phys Chem Chem Phys. 2012 May 28;14(20):7427-32. doi: 10.1039/c2cp40356k. Epub 2012 Apr 24. Phys Chem Chem Phys. 2012. PMID: 22531880
-
Triconstituent co-assembly to ordered mesostructured polymer-silica and carbon-silica nanocomposites and large-pore mesoporous carbons with high surface areas.J Am Chem Soc. 2006 Sep 6;128(35):11652-62. doi: 10.1021/ja0633518. J Am Chem Soc. 2006. PMID: 16939291
-
Exploring the confinement of polymer nanolayers into ordered mesoporous silica using advanced gas physisorption.J Colloid Interface Sci. 2020 Nov 1;579:489-507. doi: 10.1016/j.jcis.2020.05.103. Epub 2020 Jun 3. J Colloid Interface Sci. 2020. PMID: 32622098
-
Effect of Nanofillers on Tribological Properties of Polymer Nanocomposites: A Review on Recent Development.Polymers (Basel). 2021 Aug 26;13(17):2867. doi: 10.3390/polym13172867. Polymers (Basel). 2021. PMID: 34502906 Free PMC article. Review.
-
Photopolymerization Approach to Advanced Polymer Composites: Integration of Surface-Modified Nanofillers for Enhanced Properties.Adv Mater. 2024 Aug;36(33):e2400178. doi: 10.1002/adma.202400178. Epub 2024 Jun 26. Adv Mater. 2024. PMID: 38843462 Review.
Cited by
-
Porosity and surface properites of SBA-15 with grafted PNIPAAM: a water sorption calorimetry study.Langmuir. 2011 Nov 15;27(22):13838-46. doi: 10.1021/la203093u. Epub 2011 Oct 14. Langmuir. 2011. PMID: 21928772 Free PMC article.
-
Synthesis and Gas Transport Properties of Poly(2,6-dimethyl-1,4-phenylene oxide)⁻Silica Nanocomposite Membranes.Membranes (Basel). 2018 Dec 4;8(4):125. doi: 10.3390/membranes8040125. Membranes (Basel). 2018. PMID: 30518107 Free PMC article.
-
Hybrid Polymer-Silica Nanostructured Materials for Environmental Remediation.Molecules. 2023 Jun 29;28(13):5105. doi: 10.3390/molecules28135105. Molecules. 2023. PMID: 37446768 Free PMC article. Review.
-
Effect of Nanoporous Molecular Sieves TS-1 on Electrical Properties of Crosslinked Polyethylene Nanocomposites.Nanomaterials (Basel). 2024 Jun 6;14(11):985. doi: 10.3390/nano14110985. Nanomaterials (Basel). 2024. PMID: 38869610 Free PMC article.
-
Application of Silsesquioxanes in the Preparation of Polyolefin-Based Materials.Materials (Basel). 2023 Feb 24;16(5):1876. doi: 10.3390/ma16051876. Materials (Basel). 2023. PMID: 36902992 Free PMC article. Review.
References
-
- Winey K.I., Moniruzzaman M. Polymer nanocomposites containing carbon nanotubes. Macromolecules. 2006;39:5194–5205. doi: 10.1021/ma060733p. - DOI
-
- Wei L.M., Tang T., Huang B.T. Synthesis and characterization of polyethylene/clay–silica nanocomposites: a montmorillonite/silica- hybrid- supported catalyst and in situ polymerization. J. Polym. Sci. Pol. Chem. 2004;42:941–949. doi: 10.1002/pola.11053. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

