The pro-fibrotic connective tissue growth factor (CTGF/CCN2) correlates with the number of necrotic-regenerative foci in dystrophic muscle
- PMID: 28887614
- PMCID: PMC5842176
- DOI: 10.1007/s12079-017-0409-3
The pro-fibrotic connective tissue growth factor (CTGF/CCN2) correlates with the number of necrotic-regenerative foci in dystrophic muscle
Abstract
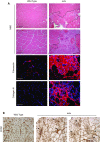
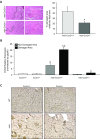
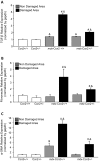
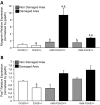
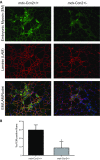
Connective tissue growth factor (CTGF/CCN2) has strong inflammatory and profibrotic activities. Its expression is enhanced in skeletal muscular dystrophies such as Duchenne muscular dystrophy (DMD), a myopathy characterized by exacerbated inflammation and fibrosis. In dystrophic tissue, necrotic-regenerative foci, myofibroblasts, newly-regenerated muscle fibers and necrosis all occur simultaneously. To determine if CCN2 is involved in the appearance of the foci, we studied their presence and characteristics in mdx mice (DMD mouse model) compared to mdx mice hemizygous for CCN2 (mdx-Ccn2+/-). We used laser capture microdissection followed by gene expression and immunofluorescence analyses to investigate fibrotic, inflammation and regeneration markers in damaged and non-damaged areas in mdx and mdx-Ccn2+/- skeletal muscle. Mdx mice foci express elevated mRNAs levels of transforming growth factor type beta, collagen, fibronectin, the myofribroblast marker α-SMA, and the myogenic transcription factor myogenin. Mdx foci also show elevated levels of MCP-1 and CD-68 positive cells, indicating that CCN2 could be inducing an inflammatory response. We found a significant reduction in the number of foci in mdx-Ccn2+/- mice muscle. Fibrotic and inflammatory markers were also decreased in these foci. We did not observe any difference in Pax7 mRNA levels, a marker for satellite cells, in mdx mice compared to mdx-Ccn2+/- mice. Thus, CCN2 appears to be involved in the fibrotic response as well as in the inflammatory response in the dystrophic skeletal muscle.
Keywords: CTGF/CCN2; Fibrosis; Inflammation; Muscular dystrophy; Necrotic-regenerative focus.
Figures
Similar articles
-
Inhibition of the angiotensin-converting enzyme decreases skeletal muscle fibrosis in dystrophic mice by a diminution in the expression and activity of connective tissue growth factor (CTGF/CCN-2).Cell Tissue Res. 2013 Jul;353(1):173-87. doi: 10.1007/s00441-013-1642-6. Epub 2013 May 15. Cell Tissue Res. 2013. PMID: 23673415
-
Reducing CTGF/CCN2 slows down mdx muscle dystrophy and improves cell therapy.Hum Mol Genet. 2013 Dec 15;22(24):4938-51. doi: 10.1093/hmg/ddt352. Epub 2013 Jul 30. Hum Mol Genet. 2013. PMID: 23904456
-
Connective tissue cells expressing fibro/adipogenic progenitor markers increase under chronic damage: relevance in fibroblast-myofibroblast differentiation and skeletal muscle fibrosis.Cell Tissue Res. 2016 Jun;364(3):647-660. doi: 10.1007/s00441-015-2343-0. Epub 2016 Jan 7. Cell Tissue Res. 2016. PMID: 26742767
-
CTGF/CCN2 from Skeletal Muscle to Nervous System: Impact on Neurodegenerative Diseases.Mol Neurobiol. 2019 Aug;56(8):5911-5916. doi: 10.1007/s12035-019-1490-9. Epub 2019 Jan 28. Mol Neurobiol. 2019. PMID: 30689195 Review.
-
HIF-hypoxia signaling in skeletal muscle physiology and fibrosis.J Cell Commun Signal. 2020 Jun;14(2):147-158. doi: 10.1007/s12079-020-00553-8. Epub 2020 Feb 22. J Cell Commun Signal. 2020. PMID: 32088838 Free PMC article. Review.
Cited by
-
Adaptations in Hippo-Yap signaling and myofibroblast fate underlie scar-free ear appendage wound healing in spiny mice.Dev Cell. 2021 Oct 11;56(19):2722-2740.e6. doi: 10.1016/j.devcel.2021.09.008. Epub 2021 Oct 4. Dev Cell. 2021. PMID: 34610329 Free PMC article.
-
Genetic manipulation of CCN2/CTGF unveils cell-specific ECM-remodeling effects in injured skeletal muscle.FASEB J. 2019 Feb;33(2):2047-2057. doi: 10.1096/fj.201800622RR. Epub 2018 Sep 14. FASEB J. 2019. PMID: 30216109 Free PMC article.
-
Imaging mass cytometry analysis of Becker muscular dystrophy muscle samples reveals different stages of muscle degeneration.Sci Rep. 2024 Feb 9;14(1):3365. doi: 10.1038/s41598-024-51906-x. Sci Rep. 2024. PMID: 38336890 Free PMC article.
-
Driving fibrosis in neuromuscular diseases: Role and regulation of Connective tissue growth factor (CCN2/CTGF).Matrix Biol Plus. 2021 Mar 6;11:100059. doi: 10.1016/j.mbplus.2021.100059. eCollection 2021 Aug. Matrix Biol Plus. 2021. PMID: 34435178 Free PMC article.
-
The linkage between inflammation and fibrosis in muscular dystrophies: The axis autotaxin-lysophosphatidic acid as a new therapeutic target?J Cell Commun Signal. 2021 Sep;15(3):317-334. doi: 10.1007/s12079-021-00610-w. Epub 2021 Mar 10. J Cell Commun Signal. 2021. PMID: 33689121 Free PMC article. Review.
References
-
- Acharyya S, Villalta SA, Bakkar N, Bupha-Intr T, Janssen PM, Carathers M, Li ZW, Beg AA, Ghosh S, Sahenk Z, Weinstein M, Gardner KL, Rafael-Fortney JA, Karin M, Tidball JG, Baldwin AS, Guttridge DC. Interplay of IKK/NF-kappaB signaling in macrophages and myofibers promotes muscle degeneration in Duchenne muscular dystrophy. J Clin Invest. 2007;117:889–901. doi: 10.1172/JCI30556. - DOI - PMC - PubMed
-
- Acuña MJ, Pessina P, Olguin H, Cabrera D, Vio CP, Bader M, Munoz-Canoves P, Santos RA, Cabello-Verrugio C, Brandan E. Restoration of muscle strength in dystrophic muscle by angiotensin-1-7 through inhibition of TGF-beta signalling. Hum Mol Genet. 2014;23:1237–1249. doi: 10.1093/hmg/ddt514. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

