Past, present, and future of Parkinson's disease: A special essay on the 200th Anniversary of the Shaking Palsy
- PMID: 28887905
- PMCID: PMC5685546
- DOI: 10.1002/mds.27115
Past, present, and future of Parkinson's disease: A special essay on the 200th Anniversary of the Shaking Palsy
Abstract
This article reviews and summarizes 200 years of Parkinson's disease. It comprises a relevant history of Dr. James Parkinson's himself and what he described accurately and what he missed from today's perspective. Parkinson's disease today is understood as a multietiological condition with uncertain etiopathogenesis. Many advances have occurred regarding pathophysiology and symptomatic treatments, but critically important issues are still pending resolution. Among the latter, the need to modify disease progression is undoubtedly a priority. In sum, this multiple-author article, prepared to commemorate the bicentenary of the shaking palsy, provides a historical state-of-the-art account of what has been achieved, the current situation, and how to progress toward resolving Parkinson's disease. © 2017 International Parkinson and Movement Disorder Society.
Keywords: 200 years anniversary; Parkinson's disease; Shaking Palsy.
© 2017 International Parkinson and Movement Disorder Society.
Conflict of interest statement
Relevant conflicts of interests/financial disclosures: Nothing to report.
Figures

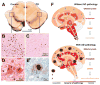

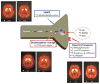

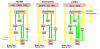


References
-
- Goldman JG, Goetz CG. History of Parkinson’s disease. Handb Clin Neurol. 2007;83:107–128. - PubMed
-
- Parkinson J, Hunter J. Hunterian Reminiscences; Being the Substance of a Course of Lectures on the Principles and Practice of Surgery, Delivered by J. Hunter, in the Year 1785. London: Sherwood, Gilbert, and Piper; 1833.
-
- Parkinson J. The Villager’s Friend and Physician. London: C Whittingham; 1804.
-
- Parkinson J. Dangerous Sports: A Tale. London: HD Symonds; 1807.
-
- Parkinson J. Some account of the effects of lightning. Mem M Soc Lon. 1789;2:490–500.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

