CD1d-Restricted pathways in hepatocytes control local natural killer T cell homeostasis and hepatic inflammation
- PMID: 28893990
- PMCID: PMC5625893
- DOI: 10.1073/pnas.1701428114
CD1d-Restricted pathways in hepatocytes control local natural killer T cell homeostasis and hepatic inflammation
Abstract
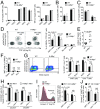
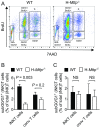
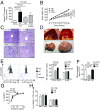
Invariant natural killer T (iNKT) cells recognize lipid antigens presented by CD1d and play a central role in regulating immunity and inflammation in peripheral tissues. However, the mechanisms which govern iNKT cell homeostasis after thymic emigration are incompletely understood. Here we demonstrate that microsomal triglyceride transfer protein (MTP), a protein involved in the transfer of lipids onto CD1d, regulates liver iNKT cell homeostasis in a manner dependent on hepatocyte CD1d. Mice with hepatocyte-specific loss of MTP exhibit defects in the function of CD1d and show increased hepatic iNKT cell numbers as a consequence of altered iNKT cell apoptosis. Similar findings were made in mice with hepatocyte-specific loss of CD1d, confirming a critical role of CD1d in this process. Moreover, increased hepatic iNKT cell abundance in the absence of MTP is associated with susceptibility to severe iNKT cell-mediated hepatitis, thus demonstrating the importance of CD1d-dependent control of liver iNKT cells in maintaining immunological homeostasis in the liver. Together, these data demonstrate an unanticipated role of parenchymal cells, as shown here for hepatocytes, in tissue-specific regulation of CD1d-restricted immunity and further suggest that alterations in lipid metabolism may affect iNKT cell homeostasis through effects on CD1d-associated lipid antigens.
Keywords: CD1d; NKT cells; hepatocyte.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

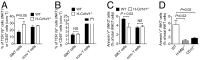
Similar articles
-
Stimulatory lipids accumulate in the mouse liver within 30 min of contact sensitization to facilitate the activation of Naïve iNKT cells in a CD1d-dependent fashion.Scand J Immunol. 2011 Jul;74(1):52-61. doi: 10.1111/j.1365-3083.2011.02540.x. Scand J Immunol. 2011. PMID: 21352253
-
Mouse and Human CD1d-Self-Lipid Complexes Are Recognized Differently by Murine Invariant Natural Killer T Cell Receptors.PLoS One. 2016 May 23;11(5):e0156114. doi: 10.1371/journal.pone.0156114. eCollection 2016. PLoS One. 2016. PMID: 27213277 Free PMC article.
-
Deletion of CD1d in Adipocytes Aggravates Adipose Tissue Inflammation and Insulin Resistance in Obesity.Diabetes. 2017 Apr;66(4):835-847. doi: 10.2337/db16-1122. Epub 2017 Jan 12. Diabetes. 2017. PMID: 28082459
-
Complex Network of NKT Cell Subsets Controls Immune Homeostasis in Liver and Gut.Front Immunol. 2018 Sep 11;9:2082. doi: 10.3389/fimmu.2018.02082. eCollection 2018. Front Immunol. 2018. PMID: 30254647 Free PMC article. Review.
-
Hiding lipid presentation: viral interference with CD1d-restricted invariant natural killer T (iNKT) cell activation.Viruses. 2012 Oct 23;4(10):2379-99. doi: 10.3390/v4102379. Viruses. 2012. PMID: 23202469 Free PMC article. Review.
Cited by
-
Type 1 invariant natural killer T cells in chronic inflammation and tissue fibrosis.Front Immunol. 2023 Sep 25;14:1260503. doi: 10.3389/fimmu.2023.1260503. eCollection 2023. Front Immunol. 2023. PMID: 37818376 Free PMC article. Review.
-
CD1-Restricted T Cells During Persistent Virus Infections: "Sympathy for the Devil".Front Immunol. 2018 Mar 19;9:545. doi: 10.3389/fimmu.2018.00545. eCollection 2018. Front Immunol. 2018. PMID: 29616036 Free PMC article. Review.
-
Acute invariant NKT cell activation triggers an immune response that drives prominent changes in iron homeostasis.Sci Rep. 2020 Dec 3;10(1):21026. doi: 10.1038/s41598-020-78037-3. Sci Rep. 2020. PMID: 33273556 Free PMC article.
-
The role of unconventional T cells in maintaining tissue homeostasis.Semin Immunol. 2022 Nov;61-64:101656. doi: 10.1016/j.smim.2022.101656. Epub 2022 Oct 25. Semin Immunol. 2022. PMID: 36306662 Free PMC article. Review. No abstract available.
-
Key features and homing properties of NK cells in the liver are shaped by activated iNKT cells.Sci Rep. 2019 Nov 8;9(1):16362. doi: 10.1038/s41598-019-52666-9. Sci Rep. 2019. PMID: 31704965 Free PMC article.
References
-
- Godfrey DI, Uldrich AP, McCluskey J, Rossjohn J, Moody DB. The burgeoning family of unconventional T cells. Nat Immunol. 2015;16:1114–1123. - PubMed
-
- Brennan PJ, Brigl M, Brenner MB. Invariant natural killer T cells: An innate activation scheme linked to diverse effector functions. Nat Rev Immunol. 2013;13:101–117. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

