A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity
- PMID: 28894149
- PMCID: PMC5593929
- DOI: 10.1038/s41467-017-00531-6
A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity
Abstract
Broad-spectrum antibiotics are frequently prescribed to children. Early childhood represents a dynamic period for the intestinal microbial ecosystem, which is readily shaped by environmental cues; antibiotic-induced disruption of this sensitive community may have long-lasting host consequences. Here we demonstrate that a single pulsed macrolide antibiotic treatment (PAT) course early in life is sufficient to lead to durable alterations to the murine intestinal microbiota, ileal gene expression, specific intestinal T-cell populations, and secretory IgA expression. A PAT-perturbed microbial community is necessary for host effects and sufficient to transfer delayed secretory IgA expression. Additionally, early-life antibiotic exposure has lasting and transferable effects on microbial community network topology. Our results indicate that a single early-life macrolide course can alter the microbiota and modulate host immune phenotypes that persist long after exposure has ceased.High or multiple doses of macrolide antibiotics, when given early in life, can perturb the metabolic and immunological development of lab mice. Here, Ruiz et al. show that even a single macrolide course, given early in life, leads to long-lasting changes in the gut microbiota and immune system of mice.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

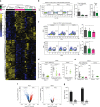

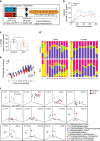
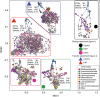
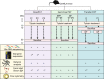
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

