Ketoprofen-loaded polymer carriers in bigel formulation: an approach to enhancing drug photostability in topical application forms
- PMID: 28894363
- PMCID: PMC5584911
- DOI: 10.2147/IJN.S140934
Ketoprofen-loaded polymer carriers in bigel formulation: an approach to enhancing drug photostability in topical application forms
Abstract
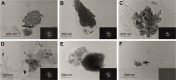
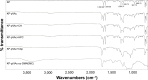
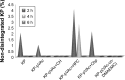
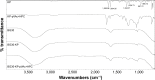
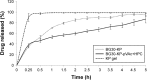
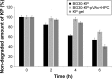
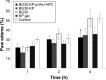
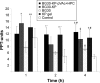
The purpose of the study was to investigate the stability and biopharmaceutical characteristics of ketoprofen, loaded in polymeric carriers, which were included into a bigel in a semisolid dosage form. The polymer carriers with in situ-included ketoprofen were obtained by emulsifier-free emulsion polymerization of the monomers in aqueous medium or a solution of the polymers used. The morphological characteristics of the carriers, the in vitro release and the photochemical stability of ketoprofen were evaluated. The model with optimal characteristics was included in a bigel formulation. The bigel was characterized in terms of pH, rheological behavior, spreadability, and in vitro drug release. Acute skin toxicity, antinociceptive activity, anti-inflammatory activity, and antihyperalgesic effects of the prepared bigel with ketoprofen-loaded polymer carrier were evaluated. The carriers of ketoprofen were characterized by a high yield and drug loading. The particle size distribution varied widely according to the polymer used, and a sustained release was provided for up to 6 hours. The polymer mixture poly(vinyl acetate) and hydroxypropyl cellulose as a drug carrier, alone or included in the bigel composition, improved the photostability of the drug compared with unprotected ketoprofen. The bigel with ketoprofen-loaded particles provided sustained release of the drug and had optimal rheological parameters. In vivo experiments on the bigel showed no skin inflammation or irritation. Four hours after its application, a well-defined analgesic, anti-inflammatory, and antihyperalgesic effect was registered. The polymer mixture of poly(vinyl acetate) and hydroxypropyl cellulose as a carrier of ketoprofen and the bigel in which it was included provided an enhanced photostability and sustained drug release.
Keywords: antihyperalgesic effect; antinociceptive activity; biphasic systems; carrageenan-induced edema; emulsifier-free radical polymerization.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures





References
-
- Bagheri H, Lhiaubet V, Montastruc JL, Chouini-Lalanne N. Photosensitivity to ketoprofen: mechanisms and pharmacoepidemiological data. Drug Saf. 2000;22(5):339–349. - PubMed
-
- Sugiura M, Hayakawa R, Xie Z, Sugiura K, Hiramoto K, Shamoto M. Experimental study on phototoxicity and the photosensitization potential of ketoprofen, suprofen, tiaprofenic acid and benzophenone and the photocross-reactivity in guinea pigs. Photodermatol Photoimmunol Photomed. 2002;18(2):82–89. - PubMed
-
- Gonçalo M. Phototoxic and photoallergic reactions. In: Johansen JD, Frosch PJ, Lepoittevin J-P, editors. Contact Dermatitis. Berlin: Springer; 2010. pp. 361–374.
-
- Matamoros V, Duhec A, Albaigés J, Bayona JM. Photodegradation of carbamazepine, ibuprofen, ketoprofen and 17 α-ethinylestradiol in fresh and seawater. Water Air soil Pollut. 2009;196(1–4):161–168.
-
- Choi YG, Lee JH, Bae IH, et al. Titanium dioxide inclusion in backing reduces the photoallergenicity of ketoprofen transdermal patch. Arch Toxicol. 2011;85(3):219–226. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

