Iron imaging reveals tumor and metastasis macrophage hemosiderin deposits in breast cancer
- PMID: 28898277
- PMCID: PMC5595304
- DOI: 10.1371/journal.pone.0184765
Iron imaging reveals tumor and metastasis macrophage hemosiderin deposits in breast cancer
Abstract
Iron-deposition is a metabolic biomarker of macrophages in both normal and pathological situations, but the presence of iron in tumor and metastasis-associated macrophages is not known. Here we mapped and quantified hemosiderin-laden macrophage (HLM) deposits in murine models of metastatic breast cancer using iron and macrophage histology, and in vivo MRI. Iron MRI detected high-iron pixel clusters in mammary tumors, lung metastasis, and brain metastasis as well as liver and spleen tissue known to contain the HLMs. Iron histology showed these regions to contain clustered macrophages identified by their common iron status and tissue-intrinsic association with other phenotypic macrophage markers. The in vivo MRI and ex vivo histological images were further processed to determine the frequencies and sizes of the iron deposits, and measure the number of HLMs in each deposit to estimate the in vivo MRI sensitivity for these cells. Hemosiderin accumulation is a macrophage biomarker and intrinsic contrast source for cellular MRI associated with the innate function of macrophages in iron metabolism systemically, and in metastatic cancer.
Conflict of interest statement
Figures
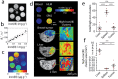
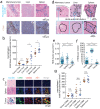


References
-
- Jaiswal S, Chao MP, Majeti R, Weissman IL. Macrophages as mediators of tumor immunosurvellance. Trends Immunol. 2010;31:212–9. doi: 10.1016/j.it.2010.04.001 - DOI - PMC - PubMed
-
- Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nat Rev Cancer. 2009;9:239–52. doi: 10.1038/nrc2618 - DOI - PMC - PubMed
-
- Lewis CE, Pollard JW. Distinct role of macrophages in different tumor microenvironments. Cancer Res. 2006;15:605–12. - PubMed
-
- Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nature medicine. 2013;19(11):1423–37. Epub 2013/11/10. doi: 10.1038/nm.3394 ; - DOI - PMC - PubMed
-
- Matovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23:549–55. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

