Taking the challenge: A protocolized approach to optimize Pneumocystis pneumonia prophylaxis in renal transplant recipients
- PMID: 28898546
- PMCID: PMC5790633
- DOI: 10.1111/ajt.14498
Taking the challenge: A protocolized approach to optimize Pneumocystis pneumonia prophylaxis in renal transplant recipients
Abstract
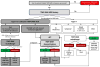
While trimethoprim-sulfamethoxazole is considered first-line therapy for Pneumocystis pneumonia prevention in renal transplant recipients, reported adverse drug reactions may limit use and increase reliance on costly and less effective alternatives, often aerosolized pentamidine. We report our experience implementing a protocolized approach to trimethoprim-sulfamethoxazole adverse drug reaction assessment and rechallenge to optimize prophylaxis in this patient cohort. We retrospectively reviewed 119 patients receiving Pneumocystis pneumonia prophylaxis prior to and after protocol implementation. Forty-two patients (35%) had 48 trimethoprim-sulfamethoxazole adverse drug reactions documented either at baseline or during the prophylaxis period, of which 83% were non-immune-mediated and 17% were immune-mediated. Significantly more patients underwent trimethoprim-sulfamethoxazole rechallenge after protocol implementation (4/22 vs 23/27; P = .0001), with no recurrence of adverse drug reactions in 74%. In those who experienced a new or recurrent reaction (26%), all were mild and self-limiting with only 1 recurrence of an immune-mediated reaction. After protocol implementation, aerosolized pentamidine-associated costs were reduced. The introduction of a standard approach to trimethoprim-sulfamethoxazole rechallenge in the context of both prior immune and non-immune-mediated reactions was safe and successful in improving the uptake of first-line Pneumocystis pneumonia prophylaxis in renal transplant recipients.
Keywords: allergy; antibiotic prophylaxis; clinical research/practice; desensitization; drug toxicity; infectious disease; kidney transplantation/nephrology; kidney transplantation: living donor.
© 2017 The American Society of Transplantation and the American Society of Transplant Surgeons.
Conflict of interest statement
Figures
References
-
- Beumont MG, Graziani A, Ubel PA, MacGregor RR. Safety of dapsone as Pneumocystis carinii pneumonia prophylaxis in human immunodeficiency virus-infected patients with allergy to trimethoprim/sulfamethoxazole. Am J Med. 1996;100(6):611–616. - PubMed
-
- Martin SI, Fishman JA, the A. S. T. Infectious Diseases Community of Practice Pneumocystis Pneumonia in Solid Organ Transplantation. Am J Transplant. 2013;13(s4):272–279. - PubMed
-
- Antibiotic Expert Group. Therapeutic guidelines: antibiotic. Version 15. Melbourne: Therapeutic Guidelines Limited; 2014.
-
- Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. [Accessed: 2/9/17];Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. Available from: http://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

