Characterization of influenza A viruses with polymorphism in PB2 residues 701 and 702
- PMID: 28900145
- PMCID: PMC5595998
- DOI: 10.1038/s41598-017-11625-y
Characterization of influenza A viruses with polymorphism in PB2 residues 701 and 702
Abstract
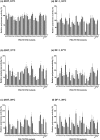
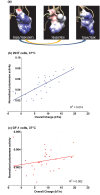
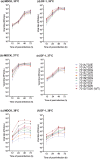
The 701 and 702 positions of influenza PB2 polymerase subunit are previously shown to have roles on host range. Limited polymorphisms at these two residues are identified in natural isolates, thereby limiting the study of their role in the polymerase. In this study, we generated 31 viable viruses by random mutagenesis at this region, indicating that these positions can tolerate a wide range of amino acids. These mutants demonstrated varying polymerase activities and viral replication rates in mammalian and avian cells. Notably, some mutants displayed enhanced polymerase activity, yet their replication kinetics were comparable to the wild-type virus. Surface electrostatic charge predication on the PB2 structural model revealed that the viral polymerase activity in mammalian cells generally increases as this region becomes more positively charged. One of the mutants (701A/702E) showed much reduced pathogenicity in mice while others had a pathogenicity similar to the wild-type level. Distinct tissue tropisms of the PB2-701/702 mutants were observed in infected chicken embryos. Overall, this study demonstrates that the PB2-701/702 region has a high degree of sequence plasticity and sequence changes in this region can alter virus phenotypes in vitro and in vivo.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

