Apolipoprotein A5 regulates intracellular triglyceride metabolism in adipocytes
- PMID: 28901468
- PMCID: PMC5865834
- DOI: 10.3892/mmr.2017.7461
Apolipoprotein A5 regulates intracellular triglyceride metabolism in adipocytes
Abstract
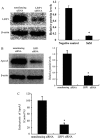
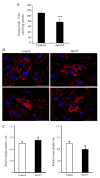
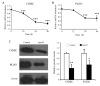
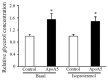
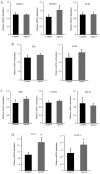
It has previously been demonstrated that apolipoprotein A5 (apoA5) can be internalized by human adipocytes and significantly decreases intracellular triglyceride content. In the present study, endocytosis of apoA5 by adipocytes under different conditions, and the underlying mechanism by which apoA5 regulates cellular triglyceride storage, was investigated. The results revealed that the apoA5 protein was detected in human subcutaneous abdominal adipose tissues. In addition, the uptake of apoA5 was attenuated in human obese adipose tissues and in cultured adipocytes with hypertrophy or insulin resistance. Low‑density lipoprotein receptor protein 1 (LRP1) knockdown in adipocytes resulted in a decrease in internalized apoA5 content, suggesting that LRP1 serves a role in apoA5 uptake. Treatment of adipocytes with apoA5 decreased the expression of the lipid droplet‑associated proteins such as cidec and perilipin. ApoA5‑treated adipocytes demonstrated an increase in lipolysis activity and expression of uncoupling protein 1, which is the molecular effector of thermogenesis in brown adipocytes. These results suggested that decreased triglyceride accumulation in adipocytes induced by apoA5 may be associated with enhanced lipolysis and energy expenditure, which may result from reduced expression of cidec and perilipin. In conclusion, the present study demonstrated a novel role of apoA5 in regulating the intracellular triglyceride metabolism of adipocytes. The results of the present study suggested that apoA5 may serve as a potential therapeutic target for the treatment of obesity and its related disorders.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

