α6 subunit-containing nicotinic receptors mediate low-dose ethanol effects on ventral tegmental area neurons and ethanol reward
- PMID: 28901722
- PMCID: PMC5849490
- DOI: 10.1111/adb.12559
α6 subunit-containing nicotinic receptors mediate low-dose ethanol effects on ventral tegmental area neurons and ethanol reward
Abstract
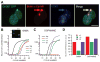
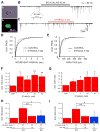
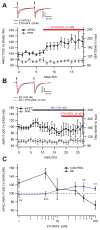
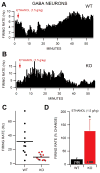
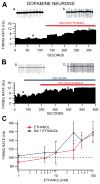
Dopamine (DA) neuron excitability is regulated by inhibitory GABAergic synaptic transmission and modulated by nicotinic acetylcholine receptors (nAChRs). The aim of this study was to evaluate the role of α6 subunit-containing nAChRs (α6*-nAChRs) in acute ethanol effects on ventral tegmental area (VTA) GABA and DA neurons. α6*-nAChRs were visualized on GABA terminals on VTA GABA neurons, and α6*-nAChR transcripts were expressed in most DA neurons, but only a minority of VTA GABA neurons from GAD67 GFP mice. Low concentrations of ethanol (1-10 mM) enhanced GABAA receptor (GABAA R)-mediated spontaneous and evoked inhibition with blockade by selective α6*-nAChR antagonist α-conotoxins (α-Ctxs) and lowered sensitivity in α6 knock-out (KO) mice. Ethanol suppression of VTA GABA neuron firing rate in wild-type mice in vivo was significantly reduced in α6 KO mice. Ethanol (5-100 mM) had no effect on optically evoked GABAA R-mediated inhibition of DA neurons, and ethanol enhancement of VTA DA neuron firing rate at high concentrations was not affected by α-Ctxs. Ethanol conditioned place preference was reduced in α6 KO mice compared with wild-type controls. Taken together, these studies indicate that relatively low concentrations of ethanol act through α6*-nAChRs on GABA terminals to enhance GABA release onto VTA GABA neurons, in turn to reduce GABA neuron firing, which may lead to VTA DA neuron disinhibition, suggesting a possible mechanism of action of alcohol and nicotine co-abuse.
Keywords: VTA; ethanol; nicotine.
© 2017 Society for the Study of Addiction.
Figures


Similar articles
-
Role of α6-Nicotinic Receptors in Alcohol-Induced GABAergic Synaptic Transmission and Plasticity to Cholinergic Interneurons in the Nucleus Accumbens.Mol Neurobiol. 2023 Jun;60(6):3113-3129. doi: 10.1007/s12035-023-03263-5. Epub 2023 Feb 18. Mol Neurobiol. 2023. PMID: 36802012 Free PMC article.
-
Nicotinic acetylcholine receptors containing the α6 subunit contribute to ethanol activation of ventral tegmental area dopaminergic neurons.Biochem Pharmacol. 2013 Oct 15;86(8):1194-200. doi: 10.1016/j.bcp.2013.06.015. Epub 2013 Jun 26. Biochem Pharmacol. 2013. PMID: 23811312 Free PMC article.
-
Functional nicotinic acetylcholine receptors containing α6 subunits are on GABAergic neuronal boutons adherent to ventral tegmental area dopamine neurons.J Neurosci. 2011 Feb 16;31(7):2537-48. doi: 10.1523/JNEUROSCI.3003-10.2011. J Neurosci. 2011. PMID: 21325521 Free PMC article.
-
Mysterious alpha6-containing nAChRs: function, pharmacology, and pathophysiology.Acta Pharmacol Sin. 2009 Jun;30(6):740-51. doi: 10.1038/aps.2009.63. Acta Pharmacol Sin. 2009. PMID: 19498417 Free PMC article. Review.
-
Cortical control of VTA function and influence on nicotine reward.Biochem Pharmacol. 2013 Oct 15;86(8):1173-80. doi: 10.1016/j.bcp.2013.07.013. Epub 2013 Aug 7. Biochem Pharmacol. 2013. PMID: 23933294 Review.
Cited by
-
Glutamate Transmission to Ventral Tegmental Area GABA Neurons Is Altered by Acute and Chronic Ethanol.Alcohol Clin Exp Res. 2018 Nov;42(11):2186-2195. doi: 10.1111/acer.13883. Epub 2018 Oct 3. Alcohol Clin Exp Res. 2018. PMID: 30204234 Free PMC article.
-
Catharanthine Modulates Mesolimbic Dopamine Transmission and Nicotine Psychomotor Effects via Inhibition of α6-Nicotinic Receptors and Dopamine Transporters.ACS Chem Neurosci. 2024 May 1;15(9):1738-1754. doi: 10.1021/acschemneuro.3c00478. Epub 2024 Apr 13. ACS Chem Neurosci. 2024. PMID: 38613458 Free PMC article.
-
The role of nicotinic acetylcholine receptors in alcohol-related behaviors.Brain Res Bull. 2020 Oct;163:135-142. doi: 10.1016/j.brainresbull.2020.07.017. Epub 2020 Jul 22. Brain Res Bull. 2020. PMID: 32707263 Free PMC article. Review.
-
A Role for GABAA Receptor β3 Subunits in Mediating Harmaline Tremor Suppression by Alcohol: Implications for Essential Tremor Therapy.Tremor Other Hyperkinet Mov (N Y). 2024 Apr 26;14:20. doi: 10.5334/tohm.834. eCollection 2024. Tremor Other Hyperkinet Mov (N Y). 2024. PMID: 38681506 Free PMC article.
-
Cocaine Directly Inhibits α6-Containing Nicotinic Acetylcholine Receptors in Human SH-EP1 Cells and Mouse VTA DA Neurons.Front Pharmacol. 2019 Feb 14;10:72. doi: 10.3389/fphar.2019.00072. eCollection 2019. Front Pharmacol. 2019. PMID: 30837868 Free PMC article.
References
-
- Allison DW, Ohran AJ, Stobbs SH, Mameli M, Valenzuela CF, Sudweeks SN, Ray AP, Henriksen SJ, Steffensen SC. Connexin-36 gap junctions mediate electrical coupling between ventral tegmental area GABA neurons. Synapse. 2006;60:20–31. - PubMed
-
- Azam L, Winzer-Serhan UH, Chen Y, Leslie FM. Expression of neuronal nicotinic acetylcholine receptor subunit mRNAs within midbrain dopamine neurons. J Comp Neurol. 2002;444:260–274. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

