Changes in the Coding and Non-coding Transcriptome and DNA Methylome that Define the Schwann Cell Repair Phenotype after Nerve Injury
- PMID: 28903050
- PMCID: PMC5608958
- DOI: 10.1016/j.celrep.2017.08.064
Changes in the Coding and Non-coding Transcriptome and DNA Methylome that Define the Schwann Cell Repair Phenotype after Nerve Injury
Abstract
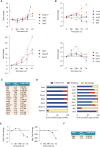
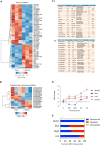
Repair Schwann cells play a critical role in orchestrating nerve repair after injury, but the cellular and molecular processes that generate them are poorly understood. Here, we perform a combined whole-genome, coding and non-coding RNA and CpG methylation study following nerve injury. We show that genes involved in the epithelial-mesenchymal transition are enriched in repair cells, and we identify several long non-coding RNAs in Schwann cells. We demonstrate that the AP-1 transcription factor C-JUN regulates the expression of certain micro RNAs in repair Schwann cells, in particular miR-21 and miR-34. Surprisingly, unlike during development, changes in CpG methylation are limited in injury, restricted to specific locations, such as enhancer regions of Schwann cell-specific genes (e.g., Nedd4l), and close to local enrichment of AP-1 motifs. These genetic and epigenomic changes broaden our mechanistic understanding of the formation of repair Schwann cell during peripheral nervous system tissue repair.
Keywords: DNA methylation; Schwann cell; c-Jun; epigenetics; long non-coding RNA; microRNA; nerve injury; nerve regeneration; repair Schwann cell.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

