Efficacy and safety of Re-Du-Ning injection in the treatment of seasonal influenza: results from a randomized, double-blinded, multicenter, oseltamivir-controlled trial
- PMID: 28903411
- PMCID: PMC5589650
- DOI: 10.18632/oncotarget.19220
Efficacy and safety of Re-Du-Ning injection in the treatment of seasonal influenza: results from a randomized, double-blinded, multicenter, oseltamivir-controlled trial
Abstract
Objective: To assess the efficacy and safety of RDNI in the treatment of seasonal influenza.
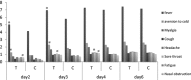
Results: 1575 participants were screened and 229 completed the study and had a RT-PCR laboratory confirmation of influenza virus infection. Fever alleviation time was 2 and 6 hours, and fever clearance time was 27 and 47 in RDNI and oseltamivir, with significant difference between two groups. Total scores of influenza symptoms descended more in RDNI than oseltamivir on day 2 and day 3. Single symptom such as fever, aversion to cold, sore throat and nasal obstruction score descended more in RDNI than oseltamivir on different days. 20 subjects used aspirin during the trial, and there was no significant difference between two groups.
Materials and methods: We conducted a randomized, double-blind, double-dummy, oseltamivir controlled clinical trial. Patients with a positive influenza rapid test diagnosis were enrolled and randomized to receive RDNI or oseltamivir. Primary outcome was the median fever alleviation and clearance time. Secondary outcomes were total 8 influenza symptom scores, the single influenza symptom score, and the frequency of aspirin usage.
Conclusions: The effect of RDNI was not worse than oseltamivir on the alleviation of influenza symptoms. RDNI was well tolerated, with no serious adverse events noted during the study period.
Keywords: Re-Du-Ning injection; clinical trial; oseltamivir; seasonal influenza.
Conflict of interest statement
CONFLICTS OF INTEREST This trial was approved by CFDA and it is under the supervision of CFDA. The aim of the trial was to expand clinical indication of drug already on the market. Everyone has no financial and personal relationships with other people or organizations that can inappropriately influence our work.
Figures



Similar articles
-
Efficacy and safety of clearing heat and detoxifying injection in the treatment of influenza: a randomized, double-blinded, placebo-controlled trial.Evid Based Complement Alternat Med. 2014;2014:151235. doi: 10.1155/2014/151235. Epub 2014 Nov 25. Evid Based Complement Alternat Med. 2014. PMID: 25506380 Free PMC article.
-
[A multicenter study of efficacy and safety of oseltamivir in treatment of naturally acquired influenza].Zhonghua Nei Ke Za Zhi. 2001 Dec;40(12):838-42. Zhonghua Nei Ke Za Zhi. 2001. PMID: 16206676 Clinical Trial. Chinese.
-
Efficacy and safety of Xiao'er Fengre Qing oral liquid versus Oseltamivir in treating pediatric influenza (wind-heat invading the defense syndrome): a multicenter, randomized, non-inferiority trial.Front Pharmacol. 2025 May 22;16:1584003. doi: 10.3389/fphar.2025.1584003. eCollection 2025. Front Pharmacol. 2025. PMID: 40474974 Free PMC article.
-
Neuraminidase inhibitors: zanamivir and oseltamivir.Ann Pharmacother. 2001 Jan;35(1):57-70. doi: 10.1345/aph.10118. Ann Pharmacother. 2001. PMID: 11197587 Review.
-
Efficacy of Lianhuaqingwen capsule compared with oseltamivir for influenza A virus infection: a meta-analysis of randomized, controlled trials.Altern Ther Health Med. 2014 Mar-Apr;20(2):25-30. Altern Ther Health Med. 2014. PMID: 24657957 Review.
Cited by
-
RDN for the treatment of influenza in children: a randomized, double-blinded, parallel-controlled clinical trial.BMC Complement Med Ther. 2023 Jul 20;23(1):255. doi: 10.1186/s12906-023-04037-1. BMC Complement Med Ther. 2023. PMID: 37474974 Free PMC article. Clinical Trial.
-
Chinese herbal injections for coronavirus disease 2019 (COVID-19): A narrative review.Integr Med Res. 2021 Dec;10(4):100778. doi: 10.1016/j.imr.2021.100778. Epub 2021 Sep 30. Integr Med Res. 2021. PMID: 34608432 Free PMC article. Review.
-
Assessing the Anti-inflammatory Mechanism of Reduning Injection by Network Pharmacology.Biomed Res Int. 2020 Dec 16;2020:6134098. doi: 10.1155/2020/6134098. eCollection 2020. Biomed Res Int. 2020. PMID: 33381562 Free PMC article.
-
Reduning Injection versus Neuraminidase Inhibitors in the Treatment of Influenza: A Systematic Review and Meta-Analysis.Chin J Integr Med. 2022 Nov;28(11):1023-1031. doi: 10.1007/s11655-022-3524-9. Epub 2022 May 4. Chin J Integr Med. 2022. PMID: 35508864 Free PMC article.
-
Efficacy and safety of ReDuNing injection as a treatment for COVID-19 and its inhibitory effect against SARS-CoV-2.J Ethnopharmacol. 2021 Oct 28;279:114367. doi: 10.1016/j.jep.2021.114367. Epub 2021 Jun 24. J Ethnopharmacol. 2021. PMID: 34174375 Free PMC article.
References
-
- WHO Global Influenza Programme A Manual for Estimating Disease Burden Associated With Seasonal Influenza. 2015 http://apps.who.int/iris/bitstream/10665/178801/1/9789241549301_eng/pdf?....
-
- Zhong NS. Respiratory Medicine. People's Medical Publishing House. 2012;24:598–604.
-
- Richman DD. Antiviral drug resistance. Antiviral Res. 2006;71:117–21. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

