Structurally Novel Antiestrogens Elicit Differential Responses from Constitutively Active Mutant Estrogen Receptors in Breast Cancer Cells and Tumors
- PMID: 28904064
- PMCID: PMC5645250
- DOI: 10.1158/0008-5472.CAN-17-1265
Structurally Novel Antiestrogens Elicit Differential Responses from Constitutively Active Mutant Estrogen Receptors in Breast Cancer Cells and Tumors
Abstract
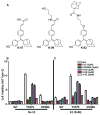
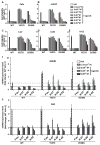
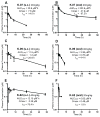
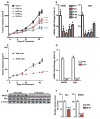
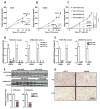
Many estrogen receptor α (ERα)-positive breast cancers develop resistance to endocrine therapy via mutation of ERs whose constitutive activation is associated with shorter patient survival. Because there is now a clinical need for new antiestrogens (AE) against these mutant ERs, we describe here our development and characterization of three chemically novel AEs that effectively suppress proliferation of breast cancer cells and tumors. Our AEs are effective against wild-type and Y537S and D538G ERs, the two most commonly occurring constitutively active ERs. The three new AEs suppressed proliferation and estrogen target gene expression in WT and mutant ER-containing cells and were more effective in D538G than in Y537S cells and tumors. Compared with WT ER, mutants exhibited approximately 10- to 20-fold lower binding affinity for AE and a reduced ability to be blocked in coactivator interaction, likely contributing to their relative resistance to inhibition by AE. Comparisons between mutant ER-containing MCF7 and T47D cells revealed that AE responses were compound, cell-type, and ERα-mutant dependent. These new ligands have favorable pharmacokinetic properties and effectively suppressed growth of WT and mutant ER-expressing tumor xenografts in NOD/SCID-γ mice after oral or subcutaneous administration; D538G tumors were more potently inhibited by AE than Y537S tumors. These studies highlight the differential responsiveness of the mutant ERs to different AEs and make clear the value of having a toolkit of AEs for treatment of endocrine therapy-resistant tumors driven by different constitutively active ERs. Cancer Res; 77(20); 5602-13. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures
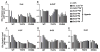
References
-
- Jeselsohn R, De Angelis C, Brown M, Schiff R. The Evolving Role of the Estrogen Receptor Mutations in Endocrine Therapy-Resistant Breast Cancer. Curr Oncol Rep. 2017;19:35. - PubMed
-
- Tryfonidis K, Zardavas D, Katzenellenbogen BS, Piccart M. Endocrine treatment in breast cancer: Cure, resistance and beyond. Cancer Treat Rev. 2016;50:68–81. - PubMed
-
- Robertson JF. Fulvestrant (Faslodex) -- how to make a good drug better. The oncologist. 2007;12:774–84. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

