Synthesis and Deployment of an Elusive Fluorovinyl Cation Equivalent: Access to Quaternary α-(1'-Fluoro)vinyl Amino Acids as Potential PLP Enzyme Inactivators
- PMID: 28906111
- PMCID: PMC6052324
- DOI: 10.1021/jacs.7b04690
Synthesis and Deployment of an Elusive Fluorovinyl Cation Equivalent: Access to Quaternary α-(1'-Fluoro)vinyl Amino Acids as Potential PLP Enzyme Inactivators
Abstract
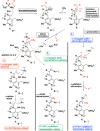
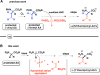
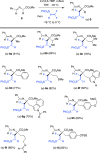
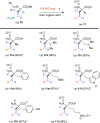
Developing specific chemical functionalities to deploy in biological environments for targeted enzyme inactivation lies at the heart of mechanism-based inhibitor development but also is central to other protein-tagging methods in modern chemical biology including activity-based protein profiling and proteolysis-targeting chimeras. We describe here a previously unknown class of potential PLP enzyme inactivators; namely, a family of quaternary, α-(1'-fluoro)vinyl amino acids, bearing the side chains of the cognate amino acids. These are obtained by the capture of suitably protected amino acid enolates with β,β-difluorovinyl phenyl sulfone, a new (1'-fluoro)vinyl cation equivalent, and an electrophile that previously eluded synthesis, capture and characterization. A significant variety of biologically relevant AA side chains are tolerated including those for alanine, valine, leucine, methionine, lysine, phenylalanine, tyrosine, and tryptophan. Following addition/elimination, the resulting transoid α-(1'-fluoro)-β-(phenylsulfonyl)vinyl AA-esters undergo smooth sulfone-stannane interchange to stereoselectively give the corresponding transoid α-(1'-fluoro)-β-(tributylstannyl)vinyl AA-esters. Protodestannylation and global deprotection then yield these sterically encumbered and densely functionalized quaternary amino acids. The α-(1'-fluoro)vinyl trigger, a potential allene-generating functionality originally proposed by Abeles, is now available in a quaternary AA context for the first time. In an initial test of this new inhibitor class, α-(1'-fluoro)vinyllysine is seen to act as a time-dependent, irreversible inactivator of lysine decarboxylase from Hafnia alvei. The enantiomers of the inhibitor could be resolved, and each is seen to give time-dependent inactivation with this enzyme. Kitz-Wilson analysis reveals similar inactivation parameters for the two antipodes, L-α-(1'-fluoro)vinyllysine (Ki = 630 ± 20 μM; t1/2 = 2.8 min) and D-α-(1'-fluoro)vinyllysine (Ki = 470 ± 30 μM; t1/2 = 3.6 min). The stage is now set for exploration of the efficacy of this trigger in other PLP-enzyme active sites.
Conflict of interest statement
Figures





Similar articles
-
Examination of the new alpha-(2'Z-fluoro)vinyl trigger with lysine decarboxylase: the absolute stereochemistry dictates the reaction course.J Am Chem Soc. 2007 Jan 17;129(2):258-9. doi: 10.1021/ja067240k. J Am Chem Soc. 2007. PMID: 17212389 Free PMC article.
-
Synthesis of quaternary amino acids bearing a (2'Z)-fluorovinyl alpha-branch: potential PLP enzyme inactivators.Org Lett. 2004 May 27;6(11):1821-4. doi: 10.1021/ol049422u. Org Lett. 2004. PMID: 15151423 Free PMC article.
-
Engineering acyclic stereocontrol in the alkylation of vinylglycine-derived dianions: asymmetric synthesis of higher alpha-vinyl amino acids.J Org Chem. 2000 May 19;65(10):2907-18. doi: 10.1021/jo9918091. J Org Chem. 2000. PMID: 10814177 Free PMC article.
-
Design of potential anticonvulsant agents: mechanistic classification of GABA aminotransferase inactivators.J Med Chem. 1989 Nov;32(11):2413-21. doi: 10.1021/jm00131a001. J Med Chem. 1989. PMID: 2681782 Review.
-
Synthesis, structure, and biological applications of α-fluorinated β-amino acids and derivatives.Chem Biodivers. 2012 Nov;9(11):2410-41. doi: 10.1002/cbdv.201200307. Chem Biodivers. 2012. PMID: 23161626 Review.
Cited by
-
Cobalt-Catalyzed Selective Unsymmetrical Dioxidation of gem-Difluoroalkenes.J Org Chem. 2020 Aug 21;85(16):10451-10465. doi: 10.1021/acs.joc.0c00415. Epub 2020 Aug 5. J Org Chem. 2020. PMID: 32697905 Free PMC article.
-
Biomacromolecule-Assisted Screening for Reaction Discovery and Catalyst Optimization.Chem Rev. 2022 Aug 24;122(16):13800-13880. doi: 10.1021/acs.chemrev.2c00213. Epub 2022 Jul 29. Chem Rev. 2022. PMID: 35904776 Free PMC article. Review.
-
Mechanism of Inactivation of Ornithine Aminotransferase by (1S,3S)-3-Amino-4-(hexafluoropropan-2-ylidenyl)cyclopentane-1-carboxylic Acid.J Am Chem Soc. 2019 Jul 10;141(27):10711-10721. doi: 10.1021/jacs.9b03254. Epub 2019 Jun 28. J Am Chem Soc. 2019. PMID: 31251613 Free PMC article.
-
Use of trifluoroacetaldehyde N-tfsylhydrazone as a trifluorodiazoethane surrogate and its synthetic applications.Nat Commun. 2019 Jan 17;10(1):284. doi: 10.1038/s41467-018-08253-z. Nat Commun. 2019. PMID: 30655536 Free PMC article.
-
α-Hydrazino Acids Inhibit Pyridoxal Phosphate-Dependent Decarboxylases via "Catalytically Correct" Ketoenamine Tautomers: A Special Motif for Chemical Biology and Drug Discovery?ACS Catal. 2025 May 2;15(10):8204-8218. doi: 10.1021/acscatal.5c00326. eCollection 2025 May 16. ACS Catal. 2025. PMID: 40401103
References
-
- Chen YC, Backus KM, Merkulova M, Yang C, Brown D, Cravatt BF, Zhang C. J Am Chem Soc. 2017;139:639–642. - PMC - PubMed
- Seki H, Xue S, Pellett S, Silhar P, Johnson EA, Janda KD. J Am Chem Soc. 2016;138:5568–5575. - PMC - PubMed
- Malcomson T, Yelekci K, Borrello MT, Ganesan A, Semina E, De Kimpe N, Mangelinckx S, Ramsay RR. FEBS J. 2015;282:3190–8. - PubMed
- Beahm BJ, Dehnert KW, Derr NL, Kuhn J, Eberhart JK, Spillmann D, Amacher SL, Bertozzi CR. Angew Chem Int Ed. 2014;53:3347–3352. - PMC - PubMed
- Lange S, Rocha-Ferreira E, Thei L, Mawjee P, Bennett K, Thompson PR, Subramanian V, Nicholas AP, Peebles D, Hristova M, Raivich G. J Neurochem. 2014;130:555–562. - PMC - PubMed
- Paul B, Das D, Ellington B, Marsh ENG. J Am Chem Soc. 2013;135:5234–5237. - PMC - PubMed
- Fuhrmann J, Subramanian V, Thompson PR. ACS Chem Biol. 2013;8:2024–2032. - PubMed
- Seeliger JC, Holsclaw CM, Schelle MW, Botyanszki Z, Gilmore SA, Tully SE, Niederweis M, Cravatt BF, Leary JA, Bertozzi CR. J Biol Chem. 2012;287:7990–8000. - PMC - PubMed
- Huang H, Chang Wc, Pai PJ, Romo A, Mansoorabadi SO, Russell DH, Liu Hw. J Am Chem Soc. 2012;134:16171–16174. - PMC - PubMed
- Shi C, Geders TW, Park SW, Wilson DJ, Boshoff HI, Abayomi O, Barry CE, Schnappinger D, Finzel BC, Aldrich CC. J Am Chem Soc. 2011;133:18194–18201. - PMC - PubMed
- Culhane JC, Wang D, Yen PM, Cole PA. J Am Chem Soc. 2010;132:3164. - PMC - PubMed
-
- Schirmeister T, Kesselring J, Jung S, Schneider TH, Weickert A, Becker J, Lee W, Bamberger D, Wich PR, Distler U, Tenzer S, Johe P, Hellmich UA, Engels B. J Am Chem Soc. 2016;138:8332–8335. - PubMed
- London N, Miller RM, Krishnan S, Uchida K, Irwin JJ, Eidam O, Gibold L, Cimermancic P, Bonnet R, Shoichet BK, Taunton J. Nat Chem Biol. 2014;10:1066–1072. - PMC - PubMed
- Noe MC, Gilbert AM. Ann Rep Med Chem. 2012;47:413–439.
- Singh J, Petter RC, Kluge AF. Curr Opin Chem Biol. 2010;14:475–480. - PubMed
- Smith AJT, Zhang X, Leach AG, Houk KN. J Med Chem. 2009;52:225–233. - PMC - PubMed
-
- Bauer RA. Drug Discovery Today. 2015;20:1061–1073. - PubMed
-
- Singh J, Petter RC, Baillie TA, Whitty A. Nat Rev Drug Discovery. 2011;10:307–317. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

