Chemical constituents and bioactivity of Formosan lauraceous plants
- PMID: 28911577
- PMCID: PMC9339549
- DOI: 10.1016/j.jfda.2015.10.008
Chemical constituents and bioactivity of Formosan lauraceous plants
Abstract
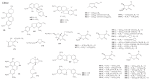
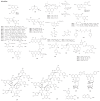
Taiwan is rich in lauraceous plants. A review of 197 references based on the chemical analysis and bioactivity of indigenous lauraceous plants carried out by native scientists from 1963 to 2014 has been compiled. About 303 new compounds and thousands of known compounds comprising alkaloids and non-alkaloids with diverse structures have been isolated or identified from indigenous plants belonging to the 11 lauraceous genera. The volatile components, however, have been excluded from this review. This review provides an overview of the past efforts of Taiwan scientists working on secondary metabolites and their bioactivity in native lauraceous plants. The potential of lauraceous plants worthy of further study is also noted. The contents will be helpful for the chemotaxonomy of Lauraceae and be of value for the development of native Formosan lauraceous plants.
Keywords: Bioactivity; Chemical constituents; Lauraceae; Review; Taiwan.
Copyright © 2016. Published by Elsevier B.V.
Figures






References
-
- Liao JC. Lauraceae in Flora of Taiwan. 2nd ed. Vol. 2. Taipei: Editorial Committee of the Flora of Taiwan; 1996. pp. 433–99.
-
- Wu YC, Liou YF, Lu ST. Antimicrobial activity of isoquinoline alkaloids and their N-oxide derivatives. Kaohsiung J Med Sci. 1988;4:336–44. - PubMed
-
- Tsai IL, Liou YF, Lu ST. Screening of isoquinoline alkaloids and their derivatives for antibacterial and antifungal activities. Kaohsiung J Med Sci. 1989;5:132–45. - PubMed
-
- Kuo YH, Lin YT. Natural research in Taiwan (I) Sci Dev. 1986;14:1223–52.
-
- Kuo YH. Natural research in Taiwan (II) Kaohsiung J Med Sci. 1989;5:360–88. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
