Long-term retention of ECM hydrogel after implantation into a sub-acute stroke cavity reduces lesion volume
- PMID: 28917705
- PMCID: PMC5653430
- DOI: 10.1016/j.actbio.2017.09.011
Long-term retention of ECM hydrogel after implantation into a sub-acute stroke cavity reduces lesion volume
Abstract
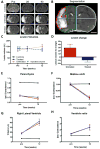
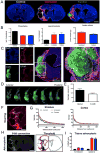
Salvaging or functional replacement of damaged tissue caused by stroke in the brain remains a major therapeutic challenge. In situ gelation and retention of a hydrogel bioscaffold composed of 8mg/mL extracellular matrix (ECM) can induce a robust invasion of cells within 24h and potentially promote a structural remodeling to replace lost tissue. Herein, we demonstrate a long-term retention of ECM hydrogel within the lesion cavity. A decrease of approximately 32% of ECM volume is observed over 12weeks. Lesion volume, as measured by magnetic resonance imaging and histology, was reduced by 28%, but a battery of behavioral tests (bilateral asymmetry test; footfault; rotameter) did not reveal a therapeutic or detrimental effect of the hydrogel. Glial scarring and peri-infarct astrocytosis were equivalent between untreated and treated animals, potentially indicating that permeation into host tissue is required to exert therapeutic effects. These results reveal a marked difference of biodegradation of ECM hydrogel in the stroke-damaged brain compared to peripheral soft tissue repair. Further exploration of these structure-function relationships is required to achieve a structural remodeling of the implanted hydrogel, as seen in peripheral tissues, to replace lost tissue and promote behavioral recovery.
Statement of significance: In situ gelation of ECM is essential for its retention within a tissue cavity. The brain is a unique environment with restricted access that necessitates image-guided delivery through a thin needle to access tissue cavities caused by stroke, as well as other conditions, such as traumatic brain injury or glioma resection. Knowledge about a brain tissue response to implanted hydrogels remains limited, especially in terms of long-term effects and potential impact on behavioral function. We here address the long-term retention of hydrogel within the brain environment, its impact on behavioral function, as well as its ability to reduce further tissue deformation caused by stroke. This study highlights considerable differences in the brain's long-term response to an ECM hydrogel compared to peripheral soft tissue. It underlines the importance of understanding the effect of the structural presence of a hydrogel within a cavity upon host brain tissue and behavioral function. As demonstrated herein, ECM hydrogel can fill a cavity long-term to reduce further progression of the cavity, while potentially serving as a reservoir for local drug or cell delivery.
Keywords: Behavior; Biodegradation; Biomaterial; Cell invasion; Extracellular matrix; Hydrogel; Implantation; Magnetic resonance imaging; Stroke; Tissue repair.
Copyright © 2017 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Figures




References
-
- Marcoli M, Agnati LF, Benedetti F, Genedani S, Guidolin D, Ferraro L, Maura G, Fuxe K. On the role of the extracellular space on the holistic behavior of the brain. Rev Neurosci. 2015;26(5):489–506. - PubMed
-
- Badylak SF, Kochupura PV, Cohen IS, Doronin SV, Saltman AE, Gilbert TW, Kelly DJ, Ignotz RA, Gaudette GR. The use of extracellular matrix as an inductive scaffold for the partial replacement of functional myocardium. Cell Transplant. 2006;15(1):S29–40. - PubMed
-
- Fallon A, Goodchild T, Wang R, Matheny RG. Remodeling of extracellular matrix patch used for carotid artery repair. The Journal of surgical research. 2012;175(1):e25–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

