Genetic engineering of human NK cells to express CXCR2 improves migration to renal cell carcinoma
- PMID: 28923105
- PMCID: PMC5604543
- DOI: 10.1186/s40425-017-0275-9
Genetic engineering of human NK cells to express CXCR2 improves migration to renal cell carcinoma
Erratum in
-
Correction to: Genetic engineering of human NK cells to express CXCR2 improves migration to renal cell carcinoma.J Immunother Cancer. 2017 Nov 6;5(1):88. doi: 10.1186/s40425-017-0292-8. J Immunother Cancer. 2017. PMID: 29110706 Free PMC article. No abstract available.
Abstract
Background: Adoptive natural killer (NK) cell transfer is being increasingly used as cancer treatment. However, clinical responses have so far been limited to patients with hematological malignancies. A potential limiting factor in patients with solid tumors is defective homing of the infused NK cells to the tumor site. Chemokines regulate the migration of leukocytes expressing corresponding chemokine receptors. Various solid tumors, including renal cell carcinoma (RCC), readily secrete ligands for the chemokine receptor CXCR2. We hypothesize that infusion of NK cells expressing high levels of the CXCR2 chemokine receptor will result in increased influx of the transferred NK cells into tumors, and improved clinical outcome in patients with cancer.
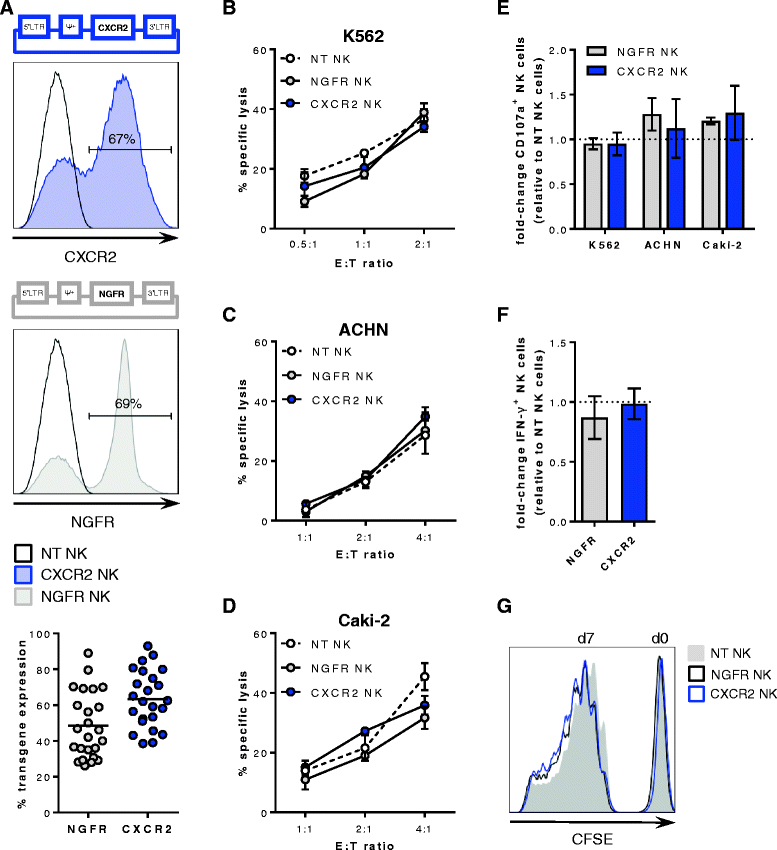
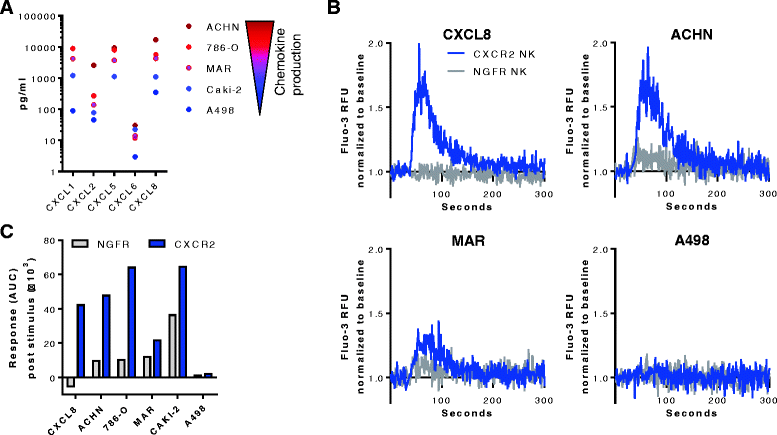
Methods: Blood and tumor biopsies from 14 primary RCC patients were assessed by flow cytometry and chemokine analysis. Primary NK cells were transduced with human CXCR2 using a retroviral system. CXCR2 receptor functionality was determined by Calcium flux and NK cell migration was evaluated in transwell assays.
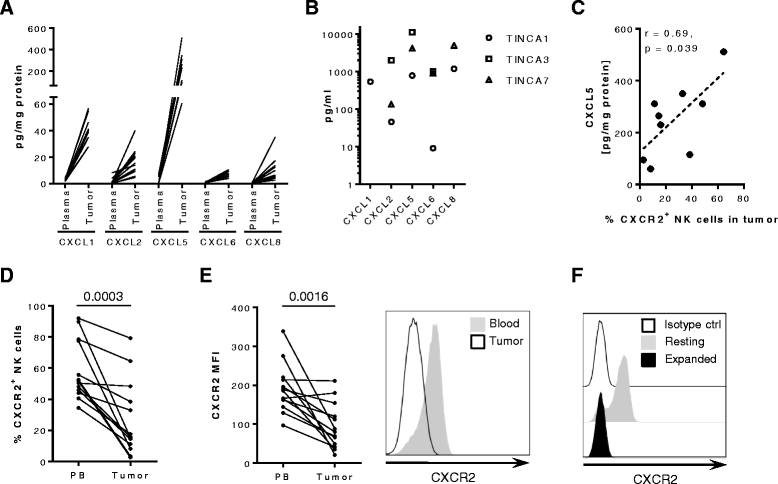
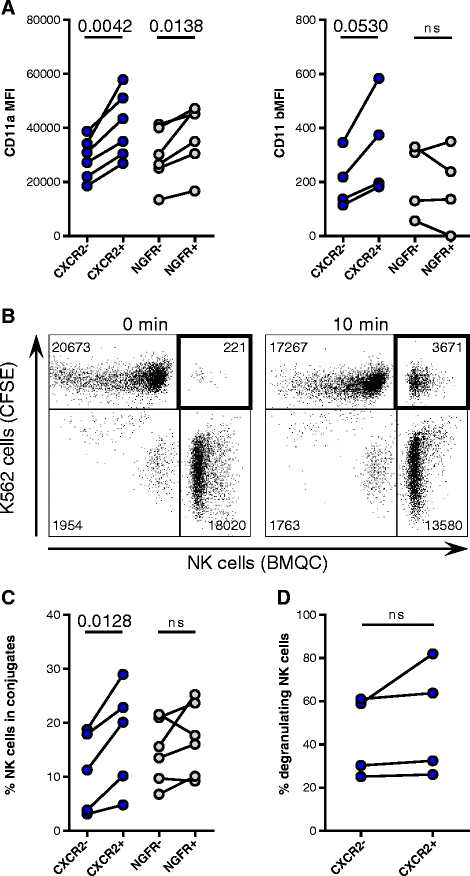
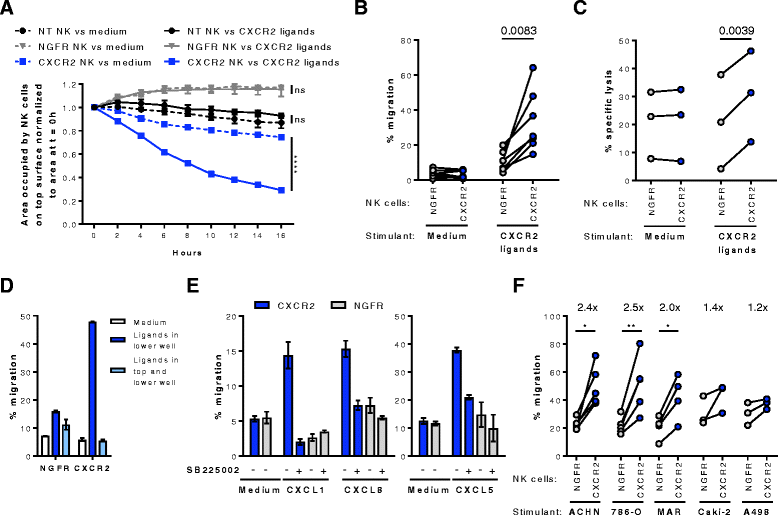
Results: We detected higher concentrations of CXCR2 ligands in tumors compared with plasma of RCC patients. In addition, CXCL5 levels correlated with the intratumoral infiltration of CXCR2-positive NK cells. However, tumor-infiltrating NK cells from RCC patients expressed lower CXCR2 compared with peripheral blood NK cells. Moreover, healthy donor NK cells rapidly lost their CXCR2 expression upon in vitro culture and expansion. Genetic modification of human primary NK cells to re-express CXCR2 improved their ability to specifically migrate along a chemokine gradient of recombinant CXCR2 ligands or RCC tumor supernatants compared with controls. The enhanced trafficking resulted in increased killing of target cells. In addition, while their functionality remained unchanged compared with control NK cells, CXCR2-transduced NK cells obtained increased adhesion properties and formed more conjugates with target cells.
Conclusions: To increase the success of NK cell-based therapies of solid tumors, it is of great importance to promote their homing to the tumor site. In this study, we show that stable engineering of human primary NK cells to express a chemokine receptor thereby enhancing their migration is a promising strategy to improve anti-tumor responses following adoptive transfer of NK cells.
Keywords: Adoptive cell therapy; CXCR2; Chemokines; NK cells; Renal cell carcinoma.
Conflict of interest statement
Ethics approval and consent to participate
The study was approved by the Regional Ethical Review Board in Stockholm (Ethical approval # 2013–570-31). All patients provided written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Viral and Nonviral Engineering of Natural Killer Cells as Emerging Adoptive Cancer Immunotherapies.J Immunol Res. 2018 Sep 17;2018:4054815. doi: 10.1155/2018/4054815. eCollection 2018. J Immunol Res. 2018. PMID: 30306093 Free PMC article. Review.
-
CXCR2-modified CAR-T cells have enhanced trafficking ability that improves treatment of hepatocellular carcinoma.Eur J Immunol. 2020 May;50(5):712-724. doi: 10.1002/eji.201948457. Epub 2020 Feb 10. Eur J Immunol. 2020. PMID: 31981231
-
Redirecting migration of T cells to chemokine secreted from tumors by genetic modification with CXCR2.Hum Gene Ther. 2002 Nov 1;13(16):1971-80. doi: 10.1089/10430340260355374. Hum Gene Ther. 2002. PMID: 12427307
-
Engineering NK-92 Cell by Upregulating CXCR2 and IL-2 Via CRISPR-Cas9 Improves Its Antitumor Effects as Cellular Immunotherapy for Human Colon Cancer.J Interferon Cytokine Res. 2021 Dec;41(12):450-460. doi: 10.1089/jir.2021.0078. J Interferon Cytokine Res. 2021. PMID: 34935484
-
Chemokine networks modulating natural killer cell trafficking to solid tumors.Cytokine Growth Factor Rev. 2021 Jun;59:36-45. doi: 10.1016/j.cytogfr.2020.12.003. Epub 2021 Jan 16. Cytokine Growth Factor Rev. 2021. PMID: 33495094 Review.
Cited by
-
Systematic improvements in lentiviral transduction of primary human natural killer cells undergoing ex vivo expansion.Mol Ther Methods Clin Dev. 2021 Jan 20;20:559-571. doi: 10.1016/j.omtm.2021.01.008. eCollection 2021 Mar 12. Mol Ther Methods Clin Dev. 2021. PMID: 33665226 Free PMC article.
-
Viral and Nonviral Engineering of Natural Killer Cells as Emerging Adoptive Cancer Immunotherapies.J Immunol Res. 2018 Sep 17;2018:4054815. doi: 10.1155/2018/4054815. eCollection 2018. J Immunol Res. 2018. PMID: 30306093 Free PMC article. Review.
-
The Role of CXC Chemokine Receptors 1-4 on Immune Cells in the Tumor Microenvironment.Front Immunol. 2018 Sep 25;9:2159. doi: 10.3389/fimmu.2018.02159. eCollection 2018. Front Immunol. 2018. PMID: 30319622 Free PMC article. Review.
-
Human CAR NK Cells: A New Non-viral Method Allowing High Efficient Transfection and Strong Tumor Cell Killing.Front Immunol. 2019 Apr 30;10:957. doi: 10.3389/fimmu.2019.00957. eCollection 2019. Front Immunol. 2019. PMID: 31114587 Free PMC article.
-
The Next Generation of Cellular Immunotherapy: Chimeric Antigen Receptor-Natural Killer Cells.Transplant Cell Ther. 2022 Oct;28(10):650-656. doi: 10.1016/j.jtct.2022.06.025. Epub 2022 Jul 3. Transplant Cell Ther. 2022. PMID: 35788086 Free PMC article. Review.
References
-
- Benson DM, Jr, Cohen AD, Jagannath S, Munshi NC, Spitzer G, Hofmeister CC, Efebera YA, Andre P, Zerbib R, Caligiuri MA. A phase I trial of the anti-KIR antibody IPH2101 and Lenalidomide in patients with relapsed/refractory multiple myeloma. Clin Cancer Res. 2015;21:4055–4061. doi: 10.1158/1078-0432.CCR-15-0304. - DOI - PMC - PubMed
-
- Miller JS, Soignier Y, Panoskaltsis-Mortari A, McNearney SA, Yun GH, Fautsch SK, McKenna D, Le C, Defor TE, Burns LJ, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood. 2005;105:3051–3057. doi: 10.1182/blood-2004-07-2974. - DOI - PubMed
-
- Geller MA, Cooley S, Judson PL, Ghebre R, Carson LF, Argenta PA, Jonson AL, Panoskaltsis-Mortari A, Curtsinger J, McKenna D, et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy. 2011;13:98–107. doi: 10.3109/14653249.2010.515582. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
