Exposure-Response Relationships for Isavuconazole in Patients with Invasive Aspergillosis and Other Filamentous Fungi
- PMID: 28923872
- PMCID: PMC5700339
- DOI: 10.1128/AAC.01034-17
Exposure-Response Relationships for Isavuconazole in Patients with Invasive Aspergillosis and Other Filamentous Fungi
Abstract
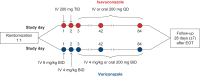
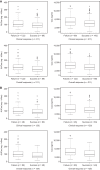
Isavuconazole, the active moiety of the water-soluble prodrug isavuconazonium sulfate, is a triazole antifungal agent for the treatment of invasive fungal infections. The purpose of this analysis was to characterize the isavuconazole exposure-response relationship for measures of efficacy and safety in patients with invasive aspergillosis and infections by other filamentous fungi from the SECURE clinical trial. Two hundred thirty-one patients who received the clinical dosing regimen and had exposure parameters were included in the analysis. The primary drug exposure parameters included were predicted trough steady-state plasma concentrations, predicted trough concentrations after 7 and 14 days of drug administration, and area under the curve estimated at steady state (AUCss). The exposure parameters were analyzed against efficacy endpoints that included all-cause mortality through day 42 in the intent-to-treat (ITT) and modified ITT populations, data review committee (DRC)-adjudicated overall response at end of treatment (EOT), and DRC-adjudicated clinical response at EOT. The safety endpoints analyzed were elevated or abnormal alanine aminotransferase, increased aspartate aminotransferase, and a combination of the two. The endpoints were analyzed using logistic regression models. No statistically significant relationship (P > 0.05) was found between isavuconazole exposure and either efficacy or safety endpoints. The lack of association between exposure and efficacy indicates that the isavuconazole exposures achieved by clinical dosing were appropriate for treating the infecting organisms in the SECURE study and that increases in alanine or aspartate aminotransferase were not related to increase in exposures. Without a clear relationship, there is no current clinical evidence for recommending routine therapeutic drug monitoring for isavuconazole.
Keywords: SECURE clinical trial; exposure-response; isavuconazole; triazole.
Copyright © 2017 Desai et al.
Figures


References
-
- Astellas Pharma Inc. US. 2015. CRESEMBA (isavuconazonium sulfate) prescribing information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/207500Orig1s000....
-
- European Medicines Agency. 2015. Cresemba (isavuconazole). http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medici... Accessed 23 February 2016.
-
- Maertens JA, Raad II, Marr KA, Patterson TF, Kontoyiannis DP, Cornely OA, Bow EJ, Rahav G, Neofytos D, Aoun M, Baddley JW, Giladi M, Heinz WJ, Herbrecht R, Hope W, Karthaus M, Lee DG, Lortholary O, Morrison VA, Oren I, Selleslag D, Shoham S, Thompson GR III, Lee M, Maher RM, Schmitt-Hoffmann AH, Zeiher B, Ullmann AJ. 2016. Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): a phase 3, randomised-controlled, non-inferiority trial. Lancet 387:760–769. doi:10.1016/S0140-6736(15)01159-9. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

