Cyclipostins and Cyclophostin analogs as promising compounds in the fight against tuberculosis
- PMID: 28924204
- PMCID: PMC5603573
- DOI: 10.1038/s41598-017-11843-4
Cyclipostins and Cyclophostin analogs as promising compounds in the fight against tuberculosis
Abstract
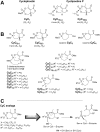
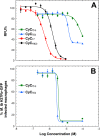
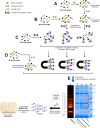
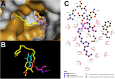
A new class of Cyclophostin and Cyclipostins (CyC) analogs have been investigated against Mycobacterium tuberculosis H37Rv (M. tb) grown either in broth medium or inside macrophages. Our compounds displayed a diversity of action by acting either on extracellular M. tb bacterial growth only, or both intracellularly on infected macrophages as well as extracellularly on bacterial growth with very low toxicity towards host macrophages. Among the eight potential CyCs identified, CyC 17 exhibited the best extracellular antitubercular activity (MIC50 = 500 nM). This compound was selected and further used in a competitive labelling/enrichment assay against the activity-based probe Desthiobiotin-FP in order to identify its putative target(s). This approach, combined with mass spectrometry, identified 23 potential candidates, most of them being serine or cysteine enzymes involved in M. tb lipid metabolism and/or in cell wall biosynthesis. Among them, Ag85A, CaeA and HsaD, have previously been reported as essential for in vitro growth of M. tb and/or survival and persistence in macrophages. Overall, our findings support the assumption that CyC 17 may thus represent a novel class of multi-target inhibitor leading to the arrest of M. tb growth through a cumulative inhibition of a large number of Ser- and Cys-containing enzymes participating in important physiological processes.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Oxadiazolone derivatives, new promising multi-target inhibitors against M. tuberculosis.Bioorg Chem. 2018 Dec;81:414-424. doi: 10.1016/j.bioorg.2018.08.025. Epub 2018 Aug 29. Bioorg Chem. 2018. PMID: 30212765
-
Cyclipostins and cyclophostin analogs inhibit the antigen 85C from Mycobacterium tuberculosis both in vitro and in vivo.J Biol Chem. 2018 Feb 23;293(8):2755-2769. doi: 10.1074/jbc.RA117.000760. Epub 2018 Jan 4. J Biol Chem. 2018. PMID: 29301937 Free PMC article.
-
Direct capture, inhibition and crystal structure of HsaD (Rv3569c) from M. tuberculosis.FEBS J. 2023 Mar;290(6):1563-1582. doi: 10.1111/febs.16645. Epub 2022 Oct 13. FEBS J. 2023. PMID: 36197115
-
Lipolytic enzymes inhibitors: A new way for antibacterial drugs discovery.Eur J Med Chem. 2021 Jan 1;209:112908. doi: 10.1016/j.ejmech.2020.112908. Epub 2020 Oct 12. Eur J Med Chem. 2021. PMID: 33071055 Review.
-
Developing pyrrole-derived antimycobacterial agents: a rational lead optimization approach.ChemMedChem. 2011 Apr 4;6(4):593-9. doi: 10.1002/cmdc.201000526. Epub 2011 Feb 21. ChemMedChem. 2011. PMID: 21341373 Review.
Cited by
-
Shifting Mycobacterial Serine Hydrolase Activity Visualized Using Multi-Layer In-Gel Activity Assays.Molecules. 2024 Jul 18;29(14):3386. doi: 10.3390/molecules29143386. Molecules. 2024. PMID: 39064965 Free PMC article.
-
Identification of covalent inhibitors that disrupt M. tuberculosis growth by targeting multiple serine hydrolases involved in lipid metabolism.Cell Chem Biol. 2022 May 19;29(5):897-909.e7. doi: 10.1016/j.chembiol.2021.08.013. Epub 2021 Oct 1. Cell Chem Biol. 2022. PMID: 34599874 Free PMC article.
-
Sequence and Structural Motifs Controlling the Broad Substrate Specificity of the Mycobacterial Hormone-Sensitive Lipase LipN.ACS Omega. 2023 Mar 30;8(14):13252-13264. doi: 10.1021/acsomega.3c00534. eCollection 2023 Apr 11. ACS Omega. 2023. PMID: 37065048 Free PMC article.
-
The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds.Molecules. 2019 Jul 16;24(14):2579. doi: 10.3390/molecules24142579. Molecules. 2019. PMID: 31315184 Free PMC article. Review.
-
Identifying Inhibitor Targets in Mycobacteria by Activity-Based Probe Profiling.Methods Mol Biol. 2025;2921:139-154. doi: 10.1007/978-1-0716-4502-4_7. Methods Mol Biol. 2025. PMID: 40515988
References
-
- WHO. Global tuberculosis report. http://www.who.int/tb/publications/global_report/en/ (2016).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

