Adipose-derived stem cell-released osteoprotegerin protects cardiomyocytes from reactive oxygen species-induced cell death
- PMID: 28931423
- PMCID: PMC5606035
- DOI: 10.1186/s13287-017-0647-6
Adipose-derived stem cell-released osteoprotegerin protects cardiomyocytes from reactive oxygen species-induced cell death
Abstract
Background: The paracrine effect is likely the major mechanism of the adipose-derived stem cell (ASC)-mediated cardioprotective effect. However, the exact composition and nature of ASC-released paracrine factors remain elusive. In the present study, we examined the effect of osteoprotegerin (OPG), a stem cell-released decoy receptor for death ligand, on the survival of cardiomyocytes exposed to oxidative stress.
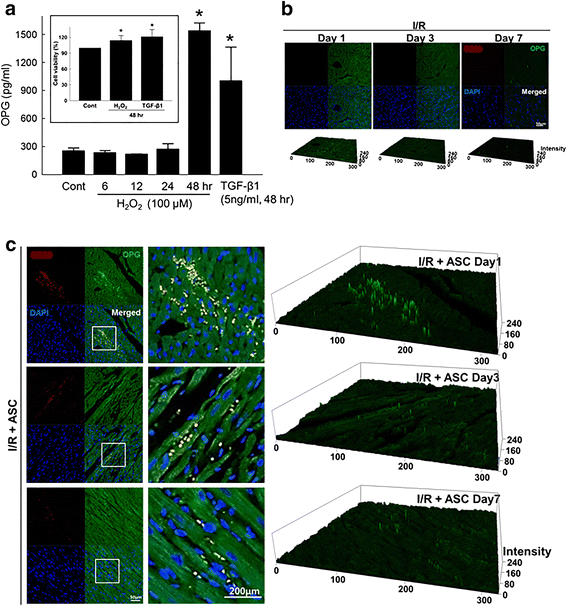
Methods: The production of OPG from ASCs under oxidative stress was determined by ELISA and immunohistochemistry. The effects of OPG and the OPG-containing conditioned media of ASCs on the survival of cardiomyocytes were determined using a cell viability assay.
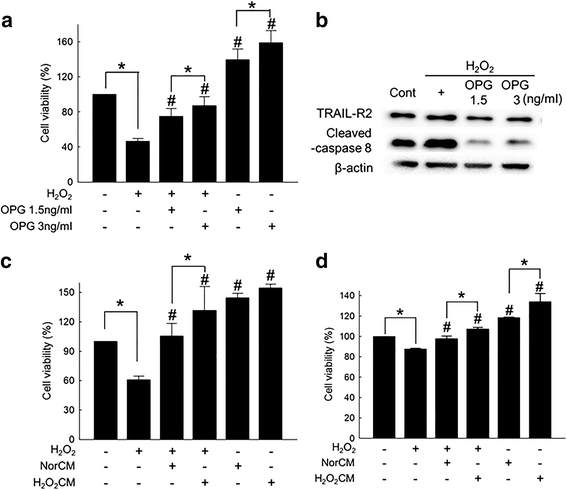
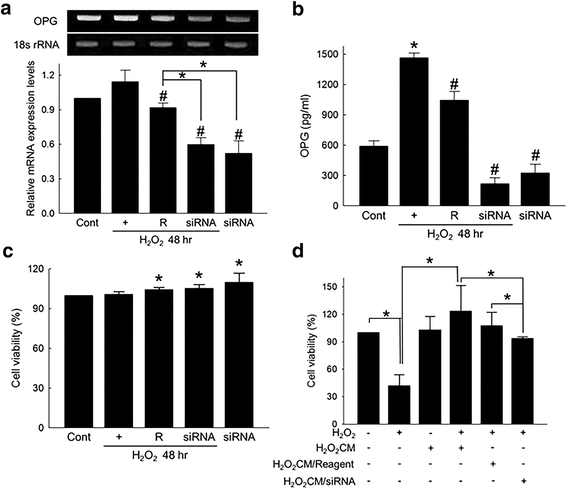
Results: Hydrogen peroxide (H2O2) significantly increased OPG production from ASCs in vitro, and OPG production from the ASCs transplanted into the ischemia-reperfusion-injured heart was also observed. OPG significantly attenuated cardiomyocyte death in vitro. OPG-containing conditioned media of ASCs also significantly protected cardiomyocytes. Delivery of siRNA specific to OPG significantly decreased the OPG production of ASCs, and also offset the protective effect of the conditioned media of ASCs.
Conclusions: Our study strongly suggests that OPG is one of the prosurvival factors released from ASCs that may contribute to the ASC-mediated cardioprotection and calls for further studies to elucidate detailed underlying mechanisms.
Keywords: Cardiomyocyte survival; Osteoprotegerin; Oxidative stress; Stem cell.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors declare their support for the publication and its contents.
Competing interests
The authors declare that they have no competing interests.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Paul A, Srivastava S, Chen G, Shum-Tim D, Prakash S. Functional assessment of adipose stem cells for xenotransplantation using myocardial infarction immunocompetent models: comparison with bone marrow stem cells. Cell Biochem Biophys. 2013;67(2):263–73. doi: 10.1007/s12013-011-9323-0. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

