(-)-Epigallocatechin-3-gallate Reduces Cigarette Smoke-Induced Airway Neutrophilic Inflammation and Mucin Hypersecretion in Rats
- PMID: 28932196
- PMCID: PMC5592236
- DOI: 10.3389/fphar.2017.00618
(-)-Epigallocatechin-3-gallate Reduces Cigarette Smoke-Induced Airway Neutrophilic Inflammation and Mucin Hypersecretion in Rats
Abstract
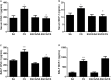
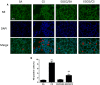
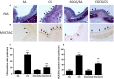
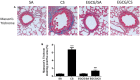
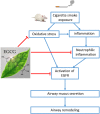
Background: Cigarette smoking is the leading cause of chronic obstructive pulmonary disease. (-)-Epigallocatechin-3-gallate (EGCG), the major catechins in Chinese green tea, has been studied for its anti-oxidative and anti-inflammatory properties in cell and animal models. In this study, we aimed to analyze the effects of EGCG on cigarette smoke (CS)-induced airway inflammation and mucus secretion in the CS-exposed rat model. Methods: Male Sprague-Dawley rats were randomly divided into either sham air (SA) or CS exposure. EGCG (50 mg/kg b.wt.) was given by oral gavage every other day in both SA and CS-exposed animals. Oxidative stress and inflammatory markers were determined in serum and/or bronchoalveolar lavage fluid by biochemical assays or ELISA. Lung morphological changes were examined by Periodic Acid-Schiff, Masson's Trichrome staining and immunohistochemical analysis. Western blot analysis was performed to explore the effects of EGCG on epidermal growth factor receptor (EGFR)-mediated signaling pathway. Results: (-)-Epigallocatechin-3-gallate treatment attenuated CS-induced oxidative stress, lung cytokine-induced neutrophil chemoattractant-1 release and neutrophil recruitment. CS exposure caused an increase in the number of goblet cells in line with MUC5AC upregulation, and increased lung collagen deposition, which were alleviated in the presence of EGCG. In addition, CS-induced phosphorylation of EGFR in rat lung was abrogated by EGCG treatment. Conclusion: (-)-Epigallocatechin-3-gallate treatment ameliorated CS-induced oxidative stress and neutrophilic inflammation, as well as airway mucus production and collagen deposition in rats. The present findings suggest that EGCG has a therapeutic effect on chronic airway inflammation and abnormal airway mucus production probably via inhibition of EGFR signaling pathway.
Keywords: (-)-epigallocatechin-3-gallate; cigarette smoke; epidermal growth factor receptor; inflammation; mucus secretion; neutrophil infiltration.
Figures


Similar articles
-
Srolo Bzhtang, a traditional Tibetan medicine formula, inhibits cigarette smoke induced airway inflammation and muc5ac hypersecretion via suppressing IL-13/STAT6 signaling pathway in rats.J Ethnopharmacol. 2019 May 10;235:424-434. doi: 10.1016/j.jep.2019.02.006. Epub 2019 Feb 5. J Ethnopharmacol. 2019. PMID: 30731182
-
Attenuation of cigarette smoke-induced airway mucus production by hydrogen-rich saline in rats.PLoS One. 2013 Dec 20;8(12):e83429. doi: 10.1371/journal.pone.0083429. eCollection 2013. PLoS One. 2013. PMID: 24376700 Free PMC article.
-
Chinese green tea ameliorates lung injury in cigarette smoke-exposed rats.Respir Med. 2009 Nov;103(11):1746-54. doi: 10.1016/j.rmed.2009.04.027. Epub 2009 May 31. Respir Med. 2009. PMID: 19487113
-
Green Tea Polyphenol (-)-Epigallocatechin-3-Gallate (EGCG): A Time for a New Player in the Treatment of Respiratory Diseases?Antioxidants (Basel). 2022 Aug 13;11(8):1566. doi: 10.3390/antiox11081566. Antioxidants (Basel). 2022. PMID: 36009285 Free PMC article. Review.
-
The efficacy of Epigallocatechin-3-gallate (green tea) in the treatment of Alzheimer's disease: an overview of pre-clinical studies and translational perspectives in clinical practice.Infect Agent Cancer. 2017 Jun 19;12:36. doi: 10.1186/s13027-017-0145-6. eCollection 2017. Infect Agent Cancer. 2017. PMID: 28642806 Free PMC article. Review.
Cited by
-
Effect of polyphenols against complications of COVID-19: current evidence and potential efficacy.Pharmacol Rep. 2024 Apr;76(2):307-327. doi: 10.1007/s43440-024-00585-6. Epub 2024 Mar 18. Pharmacol Rep. 2024. PMID: 38498260 Review.
-
Phloretin, an Apple Polyphenol, Inhibits Pathogen-Induced Mucin Overproduction.Mol Nutr Food Res. 2021 Jan;65(2):e2000658. doi: 10.1002/mnfr.202000658. Epub 2020 Dec 7. Mol Nutr Food Res. 2021. PMID: 33216464 Free PMC article.
-
Natural Products for the Management of Asthma and COPD.Handb Exp Pharmacol. 2025;287:175-205. doi: 10.1007/164_2024_709. Handb Exp Pharmacol. 2025. PMID: 38418669 Review.
-
Epigallocatechin-3-gallate ameliorates LPS-induced inflammation by inhibiting the phosphorylation of Akt and ERK signaling molecules in rat H9c2 cells.Exp Ther Med. 2020 Aug;20(2):1621-1629. doi: 10.3892/etm.2020.8827. Epub 2020 Jun 3. Exp Ther Med. 2020. PMID: 32742394 Free PMC article.
-
A Comprehensive Review of the Potential Use of Green Tea Polyphenols in the Management of COVID-19.Evid Based Complement Alternat Med. 2021 Dec 3;2021:7170736. doi: 10.1155/2021/7170736. eCollection 2021. Evid Based Complement Alternat Med. 2021. PMID: 34899956 Free PMC article. Review.
References
-
- Adler K. B., Holden-Stauffer W. J., Repine J. E. (1990). Oxygen metabolites stimulate release of high-molecular-weight glycoconjugates by cell and organ cultures of rodent respiratory epithelium via an arachidonic acid-dependent mechanism. J. Clin. Invest. 85 75–85. 10.1172/JCI114436 - DOI - PMC - PubMed
-
- ben Anes A., Fetoui H., Bchir S., ben Nasr H., Chahdoura H., Chabchoub E., et al. (2014). Increased oxidative stress and altered levels of nitric oxide and peroxynitrite in Tunisian patients with chronic obstructive pulmonary disease: correlation with disease severity and airflow obstruction. Biol. Trace Elem. Res. 161 20–31. 10.1007/s12011-014-0087-4 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

