Quantitative magnetic resonance (MR) neurography for evaluation of peripheral nerves and plexus injuries
- PMID: 28932698
- PMCID: PMC5594015
- DOI: 10.21037/qims.2017.08.01
Quantitative magnetic resonance (MR) neurography for evaluation of peripheral nerves and plexus injuries
Abstract
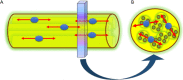
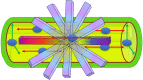
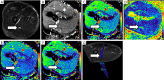
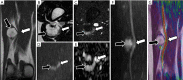
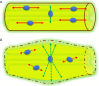
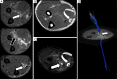
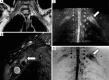
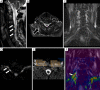
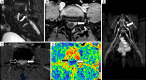
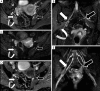
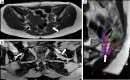
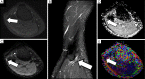
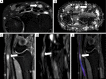
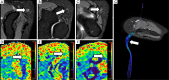
Traumatic conditions of peripheral nerves and plexus have been classically evaluated by morphological imaging techniques and electrophysiological tests. New magnetic resonance imaging (MRI) studies based on 3D fat-suppressed techniques are providing high accuracy for peripheral nerve injury evaluation from a qualitative point of view. However, these techniques do not provide quantitative information. Diffusion weighted imaging (DWI) and diffusion tensor imaging (DTI) are functional MRI techniques that are able to evaluate and quantify the movement of water molecules within different biological structures. These techniques have been successfully applied in other anatomical areas, especially in the assessment of central nervous system, and now are being imported, with promising results for peripheral nerve and plexus evaluation. DWI and DTI allow performing a qualitative and quantitative peripheral nerve analysis, providing valuable pathophysiological information about functional integrity of these structures. In the field of trauma and peripheral nerve or plexus injury, several derived parameters from DWI and DTI studies such as apparent diffusion coefficient (ADC) or fractional anisotropy (FA) among others, can be used as potential biomarkers of neural damage providing information about fiber organization, axonal flow or myelin integrity. A proper knowledge of physical basis of these techniques and their limitations is important for an optimal interpretation of the imaging findings and derived data. In this paper, a comprehensive review of the potential applications of DWI and DTI neurographic studies is performed with a focus on traumatic conditions, including main nerve entrapment syndromes in both peripheral nerves and brachial or lumbar plexus.
Keywords: Magnetic resonance imaging (MRI); diffusion MRI; diffusion tensor imaging (DTI); peripheral nerve; plexus.
Conflict of interest statement
Conflicts of Interest: The authors have no conflicts of interest to declare.
Figures




Similar articles
-
Functional MR Neurography in Evaluation of Peripheral Nerve Trauma and Postsurgical Assessment.Radiographics. 2019 Mar-Apr;39(2):427-446. doi: 10.1148/rg.2019180112. Epub 2019 Feb 8. Radiographics. 2019. PMID: 30735470 Review.
-
Feasibility of peripheral nerve MR neurography using diffusion tensor imaging adapted to skeletal muscle disease.Acta Radiol. 2018 May;59(5):560-568. doi: 10.1177/0284185117726100. Epub 2017 Aug 10. Acta Radiol. 2018. PMID: 28795588
-
Diffusion Tensor Imaging of the Brachial Plexus: A Comparison between Readout-segmented and Conventional Single-shot Echo-planar Imaging.Magn Reson Med Sci. 2019 Apr 10;18(2):150-157. doi: 10.2463/mrms.mp.2018-0004. Epub 2018 Nov 9. Magn Reson Med Sci. 2019. PMID: 30416178 Free PMC article.
-
Architectural configuration and microstructural properties of the sacral plexus: a diffusion tensor MRI and fiber tractography study.Neuroimage. 2012 Sep;62(3):1792-9. doi: 10.1016/j.neuroimage.2012.06.001. Epub 2012 Jun 13. Neuroimage. 2012. PMID: 22705377
-
Update in the evaluation of peripheral nerves by MRI, from morphological to functional neurography.Radiologia (Engl Ed). 2020 Mar-Apr;62(2):90-101. doi: 10.1016/j.rx.2019.06.005. Epub 2019 Oct 11. Radiologia (Engl Ed). 2020. PMID: 31611009 Review. English, Spanish.
Cited by
-
Identification of abnormalities in the lumbar nerve tract using diffusion-weighted magnetic resonance neurography.Eur Spine J. 2019 Apr;28(4):849-854. doi: 10.1007/s00586-018-05867-1. Epub 2019 Jan 17. Eur Spine J. 2019. PMID: 30656470
-
New insights into the evaluation of peripheral nerves lesions: a survival guide for beginners.Neuroradiology. 2022 May;64(5):875-886. doi: 10.1007/s00234-022-02916-x. Epub 2022 Feb 25. Neuroradiology. 2022. PMID: 35212785 Review.
-
Comparison of region-of-interest delineation methods for diffusion tensor imaging in patients with cervical spondylotic radiculopathy.BMC Musculoskelet Disord. 2022 Jul 15;23(1):677. doi: 10.1186/s12891-022-05639-5. BMC Musculoskelet Disord. 2022. PMID: 35840941 Free PMC article.
-
Can Magnetic Resonance Imaging Reveal Lower Motor Neuron Damage after Traumatic Spinal Cord Injury? A Scoping Review.Neurotrauma Rep. 2021 Nov 29;2(1):541-547. doi: 10.1089/neur.2021.0019. eCollection 2021. Neurotrauma Rep. 2021. PMID: 34901947 Free PMC article.
-
The Assessment and Management of Peripheral Nerve Trauma.Curr Treat Options Neurol. 2018 Jun 1;20(7):25. doi: 10.1007/s11940-018-0507-4. Curr Treat Options Neurol. 2018. PMID: 29855741 Review.
References
-
- Martín Noguerol T, Martínez Barbero JP. Advanced diffusion MRI and biomarkers in the central nervous system: a new approach. Radiologia 2017;59:273-85. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
