Withania somnifera Extract Protects Model Neurons from In Vitro Traumatic Injury
- PMID: 28933215
- PMCID: PMC5657733
- DOI: 10.1177/0963689717714320
Withania somnifera Extract Protects Model Neurons from In Vitro Traumatic Injury
Abstract
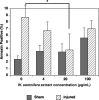
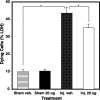
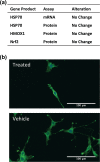
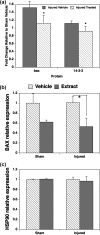
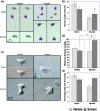
Withania somnifera has been used in traditional medicine for a variety of neural disorders. Recently, chronic neurodegenerative conditions have been shown to benefit from treatment with this extract. To evaluate the action of this extract on traumatically injured neurons, the efficacy of W. somnifera root extract as a neuroprotective agent was examined in cultured model neurons exposed to an in vitro injury system designed to mimic mild traumatic brain injury (TBI). Neuronal health was evaluated by staining with annexin V (an early, apoptotic feature) and monitoring released lactate dehydrogenase activity (a terminal cell loss parameter). Potential mechanisms underlying the observed neuroprotection were examined. Additionally, morphological changes were monitored following injury and treatment. Although no differences were found in the expression of the antioxidant transcription factor nuclear factor erythroid 2-like 2 (Nrf2) or other Nrf2-related downstream components, significant changes were seen in apoptotic signaling. Treatment with the extract resulted in an increased length of neurites projecting from the neuronal cell body after injury. W. somnifera extract treatment also resulted in reduced cell death in the model neuron TBI system. The cell death factor Bax was involved (its expression was reduced 2-fold by the treatment) and injury-induced reduction in neurite lengths and numbers was reversed by the treatment. This all indicates that W. somnifera root extract was neuroprotective and could have therapeutic potential to target factors involved in secondary injury and long-term sequelae of mild TBI.
Keywords: Ayurveda; SH-SY5Y; Withania somnifera; neurites; neuroprotection; traumatic brain injury.
Conflict of interest statement
Figures
Similar articles
-
Possible neuroprotective effect of Withania somnifera root extract against 3-nitropropionic acid-induced behavioral, biochemical, and mitochondrial dysfunction in an animal model of Huntington's disease.J Med Food. 2009 Jun;12(3):591-600. doi: 10.1089/jmf.2008.0028. J Med Food. 2009. PMID: 19627208
-
In vitro protective effects of Withania somnifera (L.) dunal root extract against hydrogen peroxide and β-amyloid(1-42)-induced cytotoxicity in differentiated PC12 cells.Phytother Res. 2010 Oct;24(10):1567-74. doi: 10.1002/ptr.3261. Phytother Res. 2010. PMID: 20680931
-
Aqueous extract from the Withania somnifera leaves as a potential anti-neuroinflammatory agent: a mechanistic study.J Neuroinflammation. 2016 Aug 22;13(1):193. doi: 10.1186/s12974-016-0650-3. J Neuroinflammation. 2016. PMID: 27550017 Free PMC article.
-
Effects of Ashwagandha (roots of Withania somnifera) on neurodegenerative diseases.Biol Pharm Bull. 2014;37(6):892-7. doi: 10.1248/bpb.b14-00022. Biol Pharm Bull. 2014. PMID: 24882401 Review.
-
Withania somnifera (L.) Dunal - Modern perspectives of an ancient Rasayana from Ayurveda.J Ethnopharmacol. 2021 Jan 10;264:113157. doi: 10.1016/j.jep.2020.113157. Epub 2020 Aug 9. J Ethnopharmacol. 2021. PMID: 32783987 Review.
Cited by
-
Neuroprotective effects of Withania somnifera in the SH-SY5Y Parkinson cell model.Heliyon. 2021 Oct 13;7(10):e08172. doi: 10.1016/j.heliyon.2021.e08172. eCollection 2021 Oct. Heliyon. 2021. PMID: 34765761 Free PMC article.
-
Gulf war toxicant-induced effects on the hippocampal dendritic arbor are reversed by treatment with a Withania somnifera extract.Front Neurosci. 2024 Feb 21;18:1368667. doi: 10.3389/fnins.2024.1368667. eCollection 2024. Front Neurosci. 2024. PMID: 38449731 Free PMC article.
-
Assessment of the Effects of Stretch-Injury on Primary Rat Microglia.Mol Neurobiol. 2021 Jul;58(7):3545-3560. doi: 10.1007/s12035-021-02362-5. Epub 2021 Mar 24. Mol Neurobiol. 2021. PMID: 33763772
-
Sodium-calcium exchanger isoform-3 targeted Withania somnifera (L.) Dunal therapeutic intervention ameliorates cognition in the 5xFAD mouse model of Alzheimer's disease.Sci Rep. 2022 Jan 27;12(1):1537. doi: 10.1038/s41598-022-05568-2. Sci Rep. 2022. PMID: 35087161 Free PMC article.
-
Role of Calcium Modulation in the Pathophysiology and Treatment of Alzheimer's Disease.Int J Mol Sci. 2023 May 22;24(10):9067. doi: 10.3390/ijms24109067. Int J Mol Sci. 2023. PMID: 37240413 Free PMC article. Review.
References
-
- Bryan-Hancock C, Harrison J. The global burden of traumatic brain injury: preliminary results from the Global Burden of Disease Project. Inj Prev. 2010;16(Suppl 1):A17.
-
- Stern RA, Riley DO, Daneshvar DH, Nowinski CJ, Cantu RC, McKee AC. Long-term consequences of repetitive brain trauma: chronic traumatic encephalopathy. PM R. 2011;3(10 Suppl 2):S460–S467. - PubMed
-
- Hatic H, Kane MJ, Saykally JN, Citron BA. Modulation of transcription factor Nrf2 in an in vitro model of traumatic brain injury. J Neurotrauma. 2012;29(6):1188–1196. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

