Chemoenzymatic Synthesis and Applications of Prokaryote-Specific UDP-Sugars
- PMID: 28935101
- PMCID: PMC6710627
- DOI: 10.1016/bs.mie.2017.06.003
Chemoenzymatic Synthesis and Applications of Prokaryote-Specific UDP-Sugars
Abstract
This method describes the chemoenzymatic synthesis of several nucleotide sugars, which are essential substrates in the biosynthesis of prokaryotic N- and O-linked glycoproteins. Protein glycosylation is now known to be widespread in prokaryotes and proceeds via sequential action of several enzymes, utilizing both common and modified prokaryote-specific sugar nucleotides. The latter, which include UDP-hexoses such as UDP-diNAc-bacillosamine (UDP-diNAcBac), UDP-diNAcAlt, and UDP-2,3-diNAcManA, are also important components of other bacterial and archaeal glycoconjugates. The ready availability of these "high-value" intermediates will enable courses of study into inhibitor screening, glycoconjugate biosynthesis pathway discovery, and unnatural carbohydrate incorporation toward metabolic engineering.
Keywords: 2,3-di-N-acetyl-glucuronic acid; 2,4-di-N-acetyl-bacillosamine; Chemoenzymatic synthesis; Glycoconjugate biosynthesis; Nucleotide-activated carbohydrates; Pseudaminic acid.
© 2017 Elsevier Inc. All rights reserved.
Figures
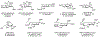
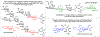
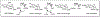
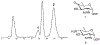
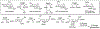
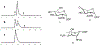
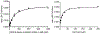
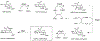
Similar articles
-
Biochemical analysis and structure determination of bacterial acetyltransferases responsible for the biosynthesis of UDP-N,N'-diacetylbacillosamine.J Biol Chem. 2013 Nov 8;288(45):32248-32260. doi: 10.1074/jbc.M113.510560. Epub 2013 Sep 24. J Biol Chem. 2013. PMID: 24064219 Free PMC article.
-
Repetitive Batch Mode Facilitates Enzymatic Synthesis of the Nucleotide Sugars UDP-Gal, UDP-GlcNAc, and UDP-GalNAc on a Multi-Gram Scale.Biotechnol J. 2019 Apr;14(4). doi: 10.1002/biot.201800386. Epub 2018 Nov 23. Biotechnol J. 2019. PMID: 30367549
-
Chemoenzymatic Synthesis of 4-Fluoro-N-Acetylhexosamine Uridine Diphosphate Donors: Chain Terminators in Glycosaminoglycan Synthesis.J Org Chem. 2017 Feb 17;82(4):2243-2248. doi: 10.1021/acs.joc.6b02929. Epub 2017 Feb 6. J Org Chem. 2017. PMID: 28128958
-
NDP-rhamnose biosynthesis and rhamnosyltransferases: building diverse glycoconjugates in nature.Biochem J. 2021 Feb 26;478(4):685-701. doi: 10.1042/BCJ20200505. Biochem J. 2021. PMID: 33599745 Review.
-
Glycans-by-design: engineering bacteria for the biosynthesis of complex glycans and glycoconjugates.Biotechnol Bioeng. 2013 Jun;110(6):1550-64. doi: 10.1002/bit.24885. Epub 2013 Mar 26. Biotechnol Bioeng. 2013. PMID: 23456823 Review.
Cited by
-
Roles of mobile genetic elements and biosynthetic gene clusters in environmental adaptation of acidophilic archaeon Ferroplasma to extreme polluted environments.Front Microbiol. 2025 Jul 31;16:1654373. doi: 10.3389/fmicb.2025.1654373. eCollection 2025. Front Microbiol. 2025. PMID: 40822404 Free PMC article.
-
Reconstitution and optimisation of the biosynthesis of bacterial sugar pseudaminic acid (Pse5Ac7Ac) enables preparative enzymatic synthesis of CMP-Pse5Ac7Ac.Sci Rep. 2021 Feb 26;11(1):4756. doi: 10.1038/s41598-021-83707-x. Sci Rep. 2021. PMID: 33637817 Free PMC article.
-
Advancements in enzyme immobilization on magnetic nanomaterials: toward sustainable industrial applications.RSC Adv. 2024 Jun 5;14(25):17946-17988. doi: 10.1039/d4ra02939a. eCollection 2024 May 28. RSC Adv. 2024. PMID: 38841394 Free PMC article. Review.
-
Metabolic Engineering of Corynebacterium glutamicum for Production of UDP-N-Acetylglucosamine.Front Bioeng Biotechnol. 2021 Sep 23;9:748510. doi: 10.3389/fbioe.2021.748510. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34631687 Free PMC article.
References
-
- Branch CL, Burton G, & Moss SF (1999). An expedient synthesis of allylic polyprenyl phosphates, Synthetic Communications, 29(15), 2639–44. doi: <Go to ISI>://000081112800013
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

