Cyclohexa-1,4-dienes in transition-metal-free ionic transfer processes
- PMID: 28936336
- PMCID: PMC5596415
- DOI: 10.1039/c7sc01657c
Cyclohexa-1,4-dienes in transition-metal-free ionic transfer processes
Abstract
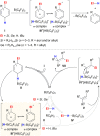
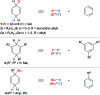
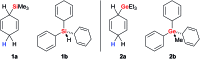
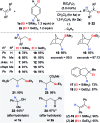
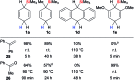
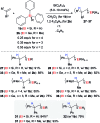
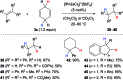
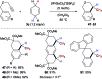
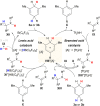
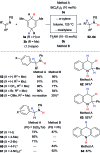
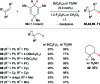
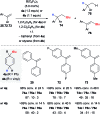
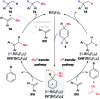
Safe- and convenient-to-handle surrogates of hazardous chemicals are always in demand. Recently introduced cyclohexa-1,4-dienes with adequate substitution fulfil this role as El+/H- equivalents in B(C6F5)3-catalysed transfer reactions of El-H to π- and σ-donors (C[double bond, length as m-dash]C/C[triple bond, length as m-dash]C and C[double bond, length as m-dash]O/C[double bond, length as m-dash]N). Surrogates of Si-H/Ge-H, H-H and even C-H bonds have been designed and successfully applied to ionic transfer hydrosilylation/hydrogermylation, hydrogenation and hydro-tert-butylation, respectively. These processes and their basic principles are summarised in this Minireview. The similarities and differences between these transfer reactions as well as the challenges associated with these transformations are discussed.
This journal is © The Royal Society of Chemistry 2017.
Figures


Similar articles
-
An air-stable, Zn2+-based catalyst for hydrosilylation of alkenes and alkynes.Org Biomol Chem. 2021 Jun 30;19(25):5544-5550. doi: 10.1039/d1ob00782c. Org Biomol Chem. 2021. PMID: 34060566
-
A motif for heteronuclear C[triple bond, length as m-dash]E (E = Si, Ge, Sn, Pb) bonding: Lewis acid-base pair strategy.Phys Chem Chem Phys. 2020 Dec 7;22(46):26720-26727. doi: 10.1039/d0cp02906h. Phys Chem Chem Phys. 2020. PMID: 33021271
-
A self-hydrosilylation of phosphanylhydrosilylalkynes promoted by B(C6F5)3? An experimental and mechanistic study.Chem Commun (Camb). 2019 Jan 29;55(10):1494-1497. doi: 10.1039/c8cc09022j. Chem Commun (Camb). 2019. PMID: 30648174
-
Metal-catalyzed silylation of sp3C-H bonds.Chem Soc Rev. 2021 Apr 26;50(8):5062-5085. doi: 10.1039/d0cs01392g. Chem Soc Rev. 2021. PMID: 33629997 Review.
-
Equilibria and mesomerism/valence tautomerism of group 4 metallocene complexes.Chem Soc Rev. 2020 Apr 7;49(7):2119-2139. doi: 10.1039/c9cs00637k. Chem Soc Rev. 2020. PMID: 32186300 Review.
Cited by
-
Lewis Acid Catalyzed Transfer Hydromethallylation for the Construction of Quaternary Carbon Centers.Angew Chem Int Ed Engl. 2019 Oct 21;58(43):15386-15389. doi: 10.1002/anie.201909852. Epub 2019 Sep 12. Angew Chem Int Ed Engl. 2019. PMID: 31449730 Free PMC article.
-
Recent advances in transition metal-free catalytic hydroelementation (E = B, Si, Ge, and Sn) of alkynes.RSC Adv. 2020 Dec 7;10(71):43539-43565. doi: 10.1039/d0ra07768b. eCollection 2020 Nov 27. RSC Adv. 2020. PMID: 35519696 Free PMC article. Review.
-
Enantioselective Organocopper-Catalyzed Hetero Diels-Alder Reaction through in Situ Oxidation of Ethers into Enol Ethers.J Am Chem Soc. 2022 Apr 13;144(14):6173-6179. doi: 10.1021/jacs.2c01656. Epub 2022 Apr 5. J Am Chem Soc. 2022. PMID: 35380438 Free PMC article.
-
FLP-Catalyzed Transfer Hydrogenation of Silyl Enol Ethers.Angew Chem Int Ed Engl. 2018 Sep 17;57(38):12356-12359. doi: 10.1002/anie.201808800. Epub 2018 Aug 24. Angew Chem Int Ed Engl. 2018. PMID: 30106498 Free PMC article.
-
Decarbonylative Transfer Hydrochlorination of Alkenes and Alkynes Based on a B(C6 F5 )3 -Initiated Grob Fragmentation.Angew Chem Int Ed Engl. 2022 Jun 13;61(24):e202203692. doi: 10.1002/anie.202203692. Epub 2022 Apr 11. Angew Chem Int Ed Engl. 2022. PMID: 35325506 Free PMC article.
References
-
- For a review, see: Wang D., Astruc D., Chem. Rev., 2015, 115 , 6621 –6686 . - PubMed
-
-
For a review of transfer hydrosilylation, see:
- Oestreich M. Angew. Chem., Int. Ed. 2016;55:494–499. - PubMed
- Simonneau A., Oestreich M. Angew. Chem., Int. Ed. 2013;52:11905–11907. - PubMed
- Simonneau A., Friebel J., Oestreich M. Eur. J. Org. Chem. 2014:2077–2083.
- Keess S., Simonneau A., Oestreich M. Organometallics. 2015;34:790–799.
- Simonneau A., Oestreich M. Nat. Chem. 2015;7:816–822. - PubMed
-
-
- Keess S., Oestreich M. Org. Lett. 2017;19:1898–1901. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

