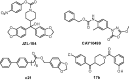
Development of terphenyl-2-methyloxazol-5(4H)-one derivatives as selective reversible MAGL inhibitors
- PMID: 28936880
- PMCID: PMC6009861
- DOI: 10.1080/14756366.2017.1375484
Development of terphenyl-2-methyloxazol-5(4H)-one derivatives as selective reversible MAGL inhibitors
Abstract
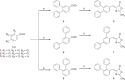
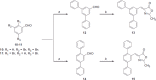
Monoacylglycerol lipase is a serine hydrolase that plays a major role in the degradation of the endocannabinoid neurotransmitter 2-arachidonoylglycerol. A wide number of MAGL inhibitors are reported in literature; however, many of them are characterised by an irreversible mechanism of action and this behavior determines an unwanted chronic MAGL inactivation, which acquires a functional antagonism of the endocannabinoid system. The possible use of reversible MAGL inhibitors has only recently been explored, due to the lack of known compounds possessing efficient reversible inhibitory activities. In this work, we report a new series of terphenyl-2-methyloxazol-5(4H)-one derivatives characterised by a reversible MAGL-inhibition mechanism. Among them, compound 20b showed to be a potent MAGL reversible inhibitor (IC50 = 348 nM) with a good MAGL/FAAH selectivity. Furthermore, this compound showed antiproliferative activities against two different cancer cell lines that overexpress MAGL.
Keywords: Monoacylglycerol lipase inhibitors; docking; endocannabinoids; molecular dynamic simulations.
Figures
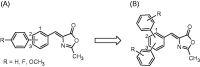

Similar articles
-
Discovery of 1,5-Diphenylpyrazole-3-Carboxamide Derivatives as Potent, Reversible, and Selective Monoacylglycerol Lipase (MAGL) Inhibitors.J Med Chem. 2018 Feb 8;61(3):1340-1354. doi: 10.1021/acs.jmedchem.7b01845. Epub 2018 Jan 22. J Med Chem. 2018. PMID: 29309142
-
4-Aryliden-2-methyloxazol-5(4H)-one as a new scaffold for selective reversible MAGL inhibitors.J Enzyme Inhib Med Chem. 2016;31(1):137-46. doi: 10.3109/14756366.2015.1010530. Epub 2015 Feb 11. J Enzyme Inhib Med Chem. 2016. PMID: 25669350
-
Identification and characterization of a new reversible MAGL inhibitor.Bioorg Med Chem. 2014 Jul 1;22(13):3285-91. doi: 10.1016/j.bmc.2014.04.057. Epub 2014 May 6. Bioorg Med Chem. 2014. PMID: 24853323
-
A patent review of Monoacylglycerol Lipase (MAGL) inhibitors (2013-2017).Expert Opin Ther Pat. 2017 Dec;27(12):1341-1351. doi: 10.1080/13543776.2018.1389899. Epub 2017 Oct 20. Expert Opin Ther Pat. 2017. PMID: 29053063 Review.
-
An updated patent review of monoacylglycerol lipase (MAGL) inhibitors (2018-present).Expert Opin Ther Pat. 2021 Feb;31(2):153-168. doi: 10.1080/13543776.2021.1841166. Epub 2020 Nov 1. Expert Opin Ther Pat. 2021. PMID: 33085920 Review.
Cited by
-
Binding investigation and preliminary optimisation of the 3-amino-1,2,4-triazin-5(2H)-one core for the development of new Fyn inhibitors.J Enzyme Inhib Med Chem. 2018 Dec;33(1):956-961. doi: 10.1080/14756366.2018.1469017. J Enzyme Inhib Med Chem. 2018. PMID: 29747534 Free PMC article.
-
The endocannabinoid system - current implications for drug development.J Intern Med. 2021 Jul;290(1):2-26. doi: 10.1111/joim.13229. Epub 2021 Jan 29. J Intern Med. 2021. PMID: 33348434 Free PMC article. Review.
-
New Chromane-Based Derivatives as Inhibitors of Mycobacterium tuberculosis Salicylate Synthase (MbtI): Preliminary Biological Evaluation and Molecular Modeling Studies.Molecules. 2018 Jun 21;23(7):1506. doi: 10.3390/molecules23071506. Molecules. 2018. PMID: 29933627 Free PMC article.
-
Antibacterial and ATP Synthesis Modulating Compounds from Salvia tingitana.J Nat Prod. 2020 Apr 24;83(4):1027-1042. doi: 10.1021/acs.jnatprod.9b01024. Epub 2020 Mar 17. J Nat Prod. 2020. PMID: 32182064 Free PMC article.
-
Aldose Reductase Differential Inhibitors in Green Tea.Biomolecules. 2020 Jul 6;10(7):1003. doi: 10.3390/biom10071003. Biomolecules. 2020. PMID: 32640594 Free PMC article.
References
-
- Di Marzo V, Petrosino S.. Endocannabinoids and the regulation of their levels in health and disease. Curr Opin Lipidol 2007;18:129–40. - PubMed
-
- Labar G, Wouters J, Lambert DM.. A review on the monoacylglycerol lipase: at the interface between fat and endocannabinoid signalling. Curr Med Chem 2010;17:2588–607. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
