Novel FXa Inhibitor Identification through Integration of Ligand- and Structure-Based Approaches
- PMID: 28937618
- PMCID: PMC6151700
- DOI: 10.3390/molecules22101588
Novel FXa Inhibitor Identification through Integration of Ligand- and Structure-Based Approaches
Abstract
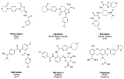
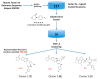
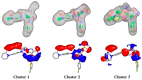
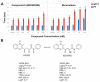
Factor Xa (FXa), a vitamin K-dependent serine protease plays a pivotal role in the coagulation cascade, one of the most interesting targets for the development of new anticoagulants. In the present work, we performed a virtual screening campaign based on ligand-based shape and electrostatic similarity search and protein-ligand docking to discover novel FXa-targeted scaffolds for further development of inhibitors. From an initial set of 260,000 compounds from the NCI Open database, 30 potential FXa inhibitors were identified and selected for in vitro biological evaluation. Compound 5 (NSC635393, 4-(3-methyl-4H-1,4-benzothiazin-2-yl)-2,4-dioxo-N-phenylbutanamide) displayed an IC50 value of 2.02 nM against human FXa. The identified compound may serve as starting point for the development of novel FXa inhibitors.
Keywords: blood coagulation cascade; enzyme inhibitors; factor Xa; protein-ligand docking; shape-based screening; virtual screening.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Innovative Three-Step Microwave-Promoted Synthesis of N-Propargyltetrahydroquinoline and 1,2,3-Triazole Derivatives as a Potential Factor Xa (FXa) Inhibitors: Drug Design, Synthesis, and Biological Evaluation.Molecules. 2020 Jan 23;25(3):491. doi: 10.3390/molecules25030491. Molecules. 2020. PMID: 31979319 Free PMC article.
-
Novel Anthranilamide-Based FXa Inhibitors: Drug Design, Synthesis and Biological Evaluation.Molecules. 2016 Apr 14;21(4):491. doi: 10.3390/molecules21040491. Molecules. 2016. PMID: 27089317 Free PMC article.
-
Identification of anthranilamide derivatives as potential factor Xa inhibitors: drug design, synthesis and biological evaluation.Eur J Med Chem. 2015 May 5;95:388-99. doi: 10.1016/j.ejmech.2015.03.052. Epub 2015 Mar 25. Eur J Med Chem. 2015. PMID: 25839438
-
Andexanet alfa: a recombinant mimetic of human factor Xa for the reversal of anticoagulant therapies.Drugs Today (Barc). 2017 May;53(5):271-282. doi: 10.1358/dot.2017.53.5.2630780. Drugs Today (Barc). 2017. PMID: 28650000 Review.
-
Surfing the Blood Coagulation Cascade: Insight into the Vital Factor Xa.Curr Med Chem. 2019;26(17):3175-3200. doi: 10.2174/0929867325666180125165340. Curr Med Chem. 2019. PMID: 29376487 Review.
Cited by
-
The Construction of a Molecular Model for the Ternary Protein Complex of Intrinsic Coagulation Pathway Factors Provides Novel Insights for the Pathogenesis of Cross-Reactive Material Positive Coagulation Factor Mutations.Int J Mol Sci. 2025 May 28;26(11):5191. doi: 10.3390/ijms26115191. Int J Mol Sci. 2025. PMID: 40508000 Free PMC article.
-
Beyond Anticoagulation: A Comprehensive Review of Non-Vitamin K Oral Anticoagulants (NOACs) in Inflammation and Protease-Activated Receptor Signaling.Int J Mol Sci. 2024 Aug 10;25(16):8727. doi: 10.3390/ijms25168727. Int J Mol Sci. 2024. PMID: 39201414 Free PMC article. Review.
-
A Structure Based Study of Selective Inhibition of Factor IXa over Factor Xa.Molecules. 2021 Sep 3;26(17):5372. doi: 10.3390/molecules26175372. Molecules. 2021. PMID: 34500804 Free PMC article.
-
Innovative Three-Step Microwave-Promoted Synthesis of N-Propargyltetrahydroquinoline and 1,2,3-Triazole Derivatives as a Potential Factor Xa (FXa) Inhibitors: Drug Design, Synthesis, and Biological Evaluation.Molecules. 2020 Jan 23;25(3):491. doi: 10.3390/molecules25030491. Molecules. 2020. PMID: 31979319 Free PMC article.
-
Convolutional Neural Network Model Based on 2D Fingerprint for Bioactivity Prediction.Int J Mol Sci. 2022 Oct 30;23(21):13230. doi: 10.3390/ijms232113230. Int J Mol Sci. 2022. PMID: 36362018 Free PMC article.
References
-
- World Health Organization (WHO) Global Status Report on Noncommunicable Diseases. WHO Publishing; Geneva, Switzerland: 2014.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

