Targeting Metabolism for Cancer Therapy
- PMID: 28938091
- PMCID: PMC5744685
- DOI: 10.1016/j.chembiol.2017.08.028
Targeting Metabolism for Cancer Therapy
Abstract
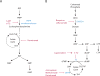
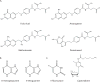
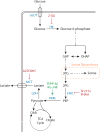
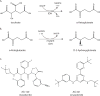
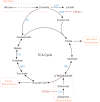
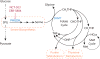
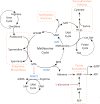
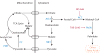
Metabolic reprogramming contributes to tumor development and introduces metabolic liabilities that can be exploited to treat cancer. Chemotherapies targeting metabolism have been effective cancer treatments for decades, and the success of these therapies demonstrates that a therapeutic window exists to target malignant metabolism. New insights into the differential metabolic dependencies of tumors have provided novel therapeutic strategies to exploit altered metabolism, some of which are being evaluated in preclinical models or clinical trials. Here, we review our current understanding of cancer metabolism and discuss how this might guide treatments targeting the metabolic requirements of tumor cells.
Keywords: cancer metabolism; cell metabolism; chemotherapy.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures

References
-
- Appel IM, den Boer ML, Meijerink JP, Veerman AJ, Reniers NC, Pieters R. Up-regulation of asparagine synthetase expression is not linked to the clinical response L-asparaginase in pediatric acute lymphoblastic leukemia. Blood. 2006;107:4244–4249. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

