Human Rab small GTPase- and class V myosin-mediated membrane tethering in a chemically defined reconstitution system
- PMID: 28939769
- PMCID: PMC5682961
- DOI: 10.1074/jbc.M117.811356
Human Rab small GTPase- and class V myosin-mediated membrane tethering in a chemically defined reconstitution system
Abstract
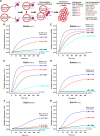
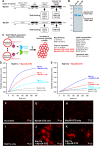
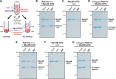
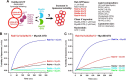
Membrane tethering is a fundamental process essential for the compartmental specificity of intracellular membrane trafficking in eukaryotic cells. Rab-family small GTPases and specific sets of Rab-interacting effector proteins, including coiled-coil tethering proteins and multisubunit tethering complexes, are reported to be responsible for membrane tethering. However, whether and how these key components directly and specifically tether subcellular membranes remains enigmatic. Using chemically defined proteoliposomal systems reconstituted with purified human Rab proteins and synthetic liposomal membranes to study the molecular basis of membrane tethering, we established here that Rab-family GTPases have a highly conserved function to directly mediate membrane tethering, even in the absence of any types of Rab effectors such as the so-called tethering proteins. Moreover, we demonstrate that membrane tethering mediated by endosomal Rab11a is drastically and selectively stimulated by its cognate Rab effectors, class V myosins (Myo5A and Myo5B), in a GTP-dependent manner. Of note, Myo5A and Myo5B exclusively recognized and cooperated with the membrane-anchored form of their cognate Rab11a to support membrane tethering mediated by trans-Rab assemblies on opposing membranes. Our findings support the novel concept that Rab-family proteins provide a bona fide membrane tether to physically and specifically link two distinct lipid bilayers of subcellular membranes. They further indicate that Rab-interacting effector proteins, including class V myosins, can regulate these Rab-mediated membrane-tethering reactions.
Keywords: Myo5; Rab; liposome; membrane reconstitution; membrane tethering; membrane trafficking; myosin; small GTPase.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures







References
-
- Bonifacino J. S., and Glick B. S. (2004) The mechanisms of vesicle budding and fusion. Cell 116, 153–166 - PubMed
-
- Jahn R., and Scheller R. H. (2006) SNAREs: Engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 7, 631–643 - PubMed
-
- Stenmark H. (2009) Rab GTPases as coordinators of vesicle traffic. Nat. Rev. Mol. Cell Biol. 10, 513–525 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

