Heterogeneous multi-compartmental hydrogel particles as synthetic cells for incompatible tandem reactions
- PMID: 28939810
- PMCID: PMC5610232
- DOI: 10.1038/s41467-017-00757-4
Heterogeneous multi-compartmental hydrogel particles as synthetic cells for incompatible tandem reactions
Abstract
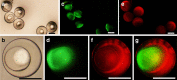
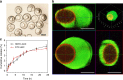
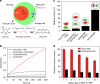
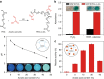
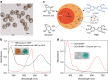
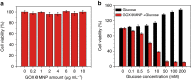
In nature, individual cells contain multiple isolated compartments in which cascade enzymatic reactions occur to form essential biological products with high efficiency. Here, we report a cell-inspired design of functional hydrogel particles with multiple compartments, in which different enzymes are spatially immobilized in distinct domains that enable engineered, one-pot, tandem reactions. The dense packing of different compartments in the hydrogel particle enables effective transportation of reactants to ensure that the products are generated with high efficiency. To demonstrate the advantages of micro-environmental modifications, we employ the copolymerization of acrylic acid, which leads to the formation of heterogeneous multi-compartmental hydrogel particles with different pH microenvironments. Upon the positional assembly of glucose oxidase and magnetic nanoparticles, these hydrogel particles are able to process a glucose-triggered, incompatible, multistep tandem reaction in one pot. Furthermore, based on the high cytotoxicity of hydroxyl radicals, a glucose-powered therapeutic strategy to kill cancer cells was approached.Cells contain isolated compartments where cascade enzymatic biochemical reactions occur to form essential biological products with high efficiency. Here the authors produce functional hydrogel particles with multiple compartments via microfluidics that contain spatially immobilized natural enzymes in distinct domains for one-pot, tandem reactions.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
Enzymatic Synthesis of Sialic Acids in Microfluidics to Overcome Cross-Inhibitions and Substrate Supply Limitations.ACS Appl Mater Interfaces. 2021 Oct 20;13(41):49433-49444. doi: 10.1021/acsami.1c12307. Epub 2021 Oct 6. ACS Appl Mater Interfaces. 2021. PMID: 34612033
-
Heterogeneous multi-compartmental DNA hydrogel particles prepared via microfluidic assembly for lymphocyte-inspired precision medicine.Nanoscale. 2021 Dec 16;13(48):20531-20540. doi: 10.1039/d1nr06594g. Nanoscale. 2021. PMID: 34859803
-
Temperature-responsive nanogel multilayers of poly(N-vinylcaprolactam) for topical drug delivery.J Colloid Interface Sci. 2017 Nov 15;506:589-602. doi: 10.1016/j.jcis.2017.07.084. Epub 2017 Jul 22. J Colloid Interface Sci. 2017. PMID: 28759859
-
Enzyme-Laden Bioactive Hydrogel for Biocatalytic Monitoring and Regulation.Acc Chem Res. 2021 Mar 2;54(5):1274-1287. doi: 10.1021/acs.accounts.0c00832. Epub 2021 Feb 11. Acc Chem Res. 2021. PMID: 33570397 Review.
-
Design of enzymatic cascade processes for the production of low-priced chemicals.Z Naturforsch C J Biosci. 2019 Feb 25;74(3-4):77-84. doi: 10.1515/znc-2018-0190. Z Naturforsch C J Biosci. 2019. PMID: 30710489 Review.
Cited by
-
Cell-inspired design of cascade catalysis system by 3D spatially separated active sites.Nat Commun. 2023 Sep 2;14(1):5338. doi: 10.1038/s41467-023-41002-5. Nat Commun. 2023. PMID: 37660124 Free PMC article.
-
Dual-signal multiplex assays based on non-tandem enzymes for simultaneous detection of multiple pathogenic bacteria.Mikrochim Acta. 2025 Aug 9;192(9):573. doi: 10.1007/s00604-025-07449-7. Mikrochim Acta. 2025. PMID: 40783448
-
Thermo-responsive aqueous two-phase system for two-level compartmentalization.Nat Commun. 2024 Aug 8;15(1):6771. doi: 10.1038/s41467-024-51043-z. Nat Commun. 2024. PMID: 39117632 Free PMC article.
-
Magnetic hydrogels with ordered structure for biomedical applications.Front Chem. 2022 Oct 11;10:1040492. doi: 10.3389/fchem.2022.1040492. eCollection 2022. Front Chem. 2022. PMID: 36304746 Free PMC article. Review.
-
Multi-compartmental MOF microreactors derived from Pickering double emulsions for chemo-enzymatic cascade catalysis.Nat Commun. 2023 Jun 3;14(1):3226. doi: 10.1038/s41467-023-38949-w. Nat Commun. 2023. PMID: 37270555 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

