Zoledronic acid augments the radiosensitivity of cancer cells through perturbing S- and M-phase cyclins and p21CIP1 expression
- PMID: 28943933
- PMCID: PMC5604142
- DOI: 10.3892/ol.2017.6710
Zoledronic acid augments the radiosensitivity of cancer cells through perturbing S- and M-phase cyclins and p21CIP1 expression
Abstract
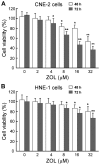
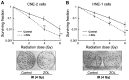
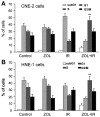
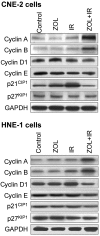
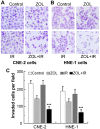
Radiotherapy and adjuvant chemotherapy have become the standard treatments for multiple types of cancer. Although cancer cells are usually sensitive to radiotherapy, metastasis and local failure still occur mainly due to developed resistance to radiotherapy. Thus, it is critical to improve therapeutics for cancer treatment. The present study demonstrated that third-generation bisphosphonate zoledronic acid (ZOL), even at a low concentration, augments the radiosensitivity of cancer cells exposed to ionizing radiation (IR) by inducing S-phase arrest and subsequently promoting apoptosis. This function of ZOL was associated with elevated levels of cyclin A and cyclin B in the S and M phases, as well as decreased p21CIP1 expression. In addition, ZOL also inhibited malignant the invasiveness of cancer cells. Notably, these effects could be enhanced concurrently with IR. The present data indicated that combined treatment with ZOL plus IR may be a novel technique to augment the radiosensitivity of cancer cells.
Keywords: radioresistance; radiosensitizing effects; zoledronic acid.
Figures
Similar articles
-
Zoledronic acid exhibits radio-sensitizing activity in human pancreatic cancer cells via inactivation of STAT3/NF-κB signaling.Onco Targets Ther. 2019 Jun 4;12:4323-4330. doi: 10.2147/OTT.S202516. eCollection 2019. Onco Targets Ther. 2019. PMID: 31239706 Free PMC article.
-
Zoledronic acid induces cell-cycle prolongation in murine lung cancer cells by perturbing cyclin and Ras expression.Anticancer Drugs. 2011 Jan;22(1):89-98. doi: 10.1097/CAD.0b013e3283400a05. Anticancer Drugs. 2011. PMID: 20926944
-
Zoledronic acid significantly enhances radiation‑induced apoptosis against human fibrosarcoma cells by inhibiting radioadaptive signaling.Int J Oncol. 2013 Feb;42(2):525-34. doi: 10.3892/ijo.2012.1735. Epub 2012 Dec 12. Int J Oncol. 2013. PMID: 23242142
-
Zoledronic acid is an effective radiosensitizer in the treatment of osteosarcoma.Oncotarget. 2016 Oct 25;7(43):70869-70880. doi: 10.18632/oncotarget.12281. Oncotarget. 2016. PMID: 27765919 Free PMC article.
-
The new bisphosphonate, Zometa (zoledronic acid), decreases skeletal complications in both osteolytic and osteoblastic lesions: a comparison to pamidronate.Cancer Invest. 2002;20 Suppl 2:45-54. doi: 10.1081/cnv-120014886. Cancer Invest. 2002. PMID: 12442349 Review.
Cited by
-
Zoledronic acid exhibits radio-sensitizing activity in human pancreatic cancer cells via inactivation of STAT3/NF-κB signaling.Onco Targets Ther. 2019 Jun 4;12:4323-4330. doi: 10.2147/OTT.S202516. eCollection 2019. Onco Targets Ther. 2019. PMID: 31239706 Free PMC article.
-
The Effect of Polydeoxyribonucleotide Extracted from Salmon Sperm on the Restoration of Bisphosphonate-Related Osteonecrosis of the Jaw.Mar Drugs. 2019 Jan 11;17(1):51. doi: 10.3390/md17010051. Mar Drugs. 2019. PMID: 30641942 Free PMC article.
-
Multiple functions of p21 in cancer radiotherapy.J Cancer Res Clin Oncol. 2021 Apr;147(4):987-1006. doi: 10.1007/s00432-021-03529-2. Epub 2021 Feb 5. J Cancer Res Clin Oncol. 2021. PMID: 33547489 Free PMC article. Review.
-
Carbon-Ion Beam Irradiation Alone or in Combination with Zoledronic acid Effectively Kills Osteosarcoma Cells.Cancers (Basel). 2020 Mar 16;12(3):698. doi: 10.3390/cancers12030698. Cancers (Basel). 2020. PMID: 32187978 Free PMC article.
References
-
- Minsky BD, Pajak TF, Ginsberg RJ, Pisansky TM, Martenson J, Komaki R, Okawara G, Rosenthal SA, Kelsen DP. INT 0123 (radiation therapy oncology group 94-05) phase III trial of combined-modality therapy for esophageal cancer: High-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20:1167–1174. doi: 10.1200/JCO.20.5.1167. - DOI - PubMed
-
- Al-Sarraf M, LeBlanc M, Giri PG, Fu KK, Cooper J, Vuong T, Forastiere AA, Adams G, Sakr WA, Schuller DE, Ensley JF. Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: Phase III randomized Intergroup study 0099. J Clin Oncol. 1998;16:1310–1317. doi: 10.1200/JCO.1998.16.4.1310. - DOI - PubMed
-
- Mao YP, Zhou GQ, Liu LZ, Guo R, Sun Y, Li L, Lin AH, Zeng MS, Kang TB, Jia WH, et al. Comparison of radiological and clinical features of temporal lobe necrosis in nasopharyngeal carcinoma patients treated with 2D radiotherapy or intensity-modulated radiotherapy. Br J Cancer. 2014;110:2633–2639. doi: 10.1038/bjc.2014.243. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
