An Update on Treatment of Pediatric Chronic Non-Infectious Uveitis
- PMID: 28944162
- PMCID: PMC5604477
- DOI: 10.1007/s40674-017-0057-z
An Update on Treatment of Pediatric Chronic Non-Infectious Uveitis
Abstract
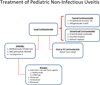
There are no standardized treatment protocols for pediatric non-infectious uveitis. Topical corticosteroids are the typical first-line agent, although systemic corticosteroids are used in intermediate, posterior and panuveitic uveitis. Corticosteroids are not considered to be long-term therapy due to potential ocular and systemic side effects. In children with severe and/or refractory uveitis, timely management with higher dose disease-modifying antirheumatic drugs (DMARDs) and biologic agents is important. Increased doses earlier in the disease course may lead to improved disease control and better visual outcomes. In general, methotrexate is the usual first-line steroid-sparing agent and given as a subcutaneous weekly injection at >0.5 mg/kg/dose or 10-15 mg/m2 due to better bioavailability. Other DMARDs, for instance mycophenolate, azathioprine, and cyclosporine are less common treatments for pediatric uveitis. Anti-tumor necrosis factor-alpha agents, primarily infliximab and adalimumab are used as second line agents in children refractory to methotrexate, or as first-line treatment in those with severe complicated disease at presentation. Infliximab may be given at a minimum of 7.5 mg/kg/dose every 4 weeks after loading doses, up to 20 mg/kg/dose. Adalimumab may be given up to 20 or 40 mg weekly. In children who fail anti-tumor necrosis factor-alpha agents, develop anti-tumor necrosis factor-alpha antibodies, experience adverse effects, or have difficulty with tolerance, there is less data available regarding subsequent treatment. Promising results have been noted with tocilizumab infusions every 2-4 weeks, abatacept monthly infusions and rituximab.
Keywords: Adalimumab; Infliximab; Juvenile Idiopathic Arthritis; Methotrexate; Pediatric uveitis; Uveitis.
Conflict of interest statement
Compliance with Ethics Guidelines Conflict of Interest Arjun B. Sood, MD and Sheila T. Angeles-Han, MD, MSc declare that they have no conflict of interest. Dr. Angeles-Han was supported by Award Number K23EY021760 from the National Eye Institute and also by a grant from the American College of Rheumatology Research and Education Foundation Career Development Bridge Funding Award. However, these did not support this study.
Figures
Similar articles
-
The effect of anti-tumor necrosis factor alpha agents on the outcome in pediatric uveitis of diverse etiologies.Graefes Arch Clin Exp Ophthalmol. 2018 Apr;256(4):801-808. doi: 10.1007/s00417-018-3928-6. Epub 2018 Feb 18. Graefes Arch Clin Exp Ophthalmol. 2018. PMID: 29455252
-
PAPA Syndrome: Challenges in Achieving Long-Term Remission.Acta Dermatovenerol Croat. 2023 Nov;31(2):106-109. Acta Dermatovenerol Croat. 2023. PMID: 38006373
-
Update on biologic therapies for juvenile idiopathic arthritis-associated uveitis.Ann Eye Sci. 2021 Jun;6:19. doi: 10.21037/aes-2019-dmu-10. Epub 2021 Jun 15. Ann Eye Sci. 2021. PMID: 34131629 Free PMC article.
-
Update of the evidence based, interdisciplinary guideline for anti-inflammatory treatment of uveitis associated with juvenile idiopathic arthritis.Semin Arthritis Rheum. 2019 Aug;49(1):43-55. doi: 10.1016/j.semarthrit.2018.11.004. Epub 2018 Dec 4. Semin Arthritis Rheum. 2019. PMID: 30595409 Review.
-
Treatment of Juvenile Idiopathic Arthritis-Associated Uveitis.Turk J Ophthalmol. 2016 Apr;46(2):77-82. doi: 10.4274/tjo.09581. Epub 2016 Apr 5. Turk J Ophthalmol. 2016. PMID: 27800265 Free PMC article. Review.
Cited by
-
Efficacy and Safety of Adalimumab in Pediatric Non-infectious Non-anterior Uveitis: Real-life Experience From the International AIDA Network Uveitis Registry.Ophthalmol Ther. 2023 Aug;12(4):1957-1971. doi: 10.1007/s40123-023-00712-1. Epub 2023 May 11. Ophthalmol Ther. 2023. PMID: 37166627 Free PMC article.
-
Visual Function and Intraocular Pressure Outcomes Following Surgical Intervention in Pediatric Patients with Inflammatory Glaucoma.Ocul Immunol Inflamm. 2025 May 23:1-7. doi: 10.1080/09273948.2025.2508408. Online ahead of print. Ocul Immunol Inflamm. 2025. PMID: 40408559
-
Pediatric peripapillary choroidal neovascularization secondary to ocular sarcoidosis: a long-term follow-up case.BMC Ophthalmol. 2025 Jul 23;25(1):422. doi: 10.1186/s12886-025-04266-7. BMC Ophthalmol. 2025. PMID: 40702433 Free PMC article.
-
Experience of using adalimumab in treating sight-threatening paediatric or adolescent Behcet's disease-related uveitis.J Ophthalmic Inflamm Infect. 2019 Jul 31;9(1):14. doi: 10.1186/s12348-019-0181-z. J Ophthalmic Inflamm Infect. 2019. PMID: 31367810 Free PMC article.
-
The Efficacy of Adalimumab in Children with Chronic Non-infectious Posterior Uveitis and Panuveitis: A Retrospective Cohort Study.Ophthalmol Ther. 2024 May;13(5):1239-1253. doi: 10.1007/s40123-024-00884-4. Epub 2024 Mar 18. Ophthalmol Ther. 2024. PMID: 38498278 Free PMC article.
References
-
- Simonini G, Cantarini L, Bresci C, Lorusso M, Galeazzi M, Cimaz R. Current therapeutic approaches to autoimmune chronic uveitis in children. Autoimmunity reviews. 2010;9(10):674–683. - PubMed
-
- Dhoot DS, Martin DF, Srivastava SK. Pediatric infectious posterior uveitis. International ophthalmology clinics. 2011;51(1):113–128. - PubMed
-
- Kim SJ. Diagnosis and management of noninfectious pediatric uveitis. International ophthalmology clinics. 2011;51(1):129–145. - PubMed
-
- Levy-Clarke GA, Nussenblatt RB, Smith JA. Management of chronic pediatric uveitis. Current opinion in ophthalmology. 2005;16(5):281–288. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
