Analysis of proteomic differences between liquefied after-cataracts and normal lenses using two-dimensional gel electrophoresis and mass spectrometry
- PMID: 28944190
- PMCID: PMC5596216
- DOI: 10.18240/ijo.2017.09.02
Analysis of proteomic differences between liquefied after-cataracts and normal lenses using two-dimensional gel electrophoresis and mass spectrometry
Abstract
Aim: To analyze and identify the proteomic differences between liquefied after-cataracts and normal lenses by means of liquefied chromatography-tandem mass spectrometry (LC-MS/MS).
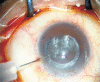
Methods: Three normal lenses and three liquefied after-cataracts were exposed to depolymerizing reagents to extract the total proteins. Protein concentrations were separated using two-dimensional gel electrophoresis (2-DE). The digitized images obtained with a GS-800 scanner were then analyzed with PDQuest7.0 software to detect the differentially-expressed protein spots. These protein spots were cut from the gel using a proteome work spot cutter and subjected to in-gel digestion with trypsin. The digested peptide separation was conducted by LC-MS/MS.
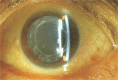
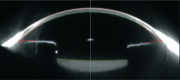
Results: The 2-DE maps showed that lens proteins were in a pH range of 3-10 with a relative molecular weight of 21-70 kD. The relative molecular weight of the more abundant proteins was localized at 25-50 kD, and the isoelectric points were found to lie between PI 4-9. The maps also showed that the protein level within the liquefied after-cataracts was at 29 points and significantly lower than in normal lenses. The 29 points were identified by LC-MS/MS, and ten of these proteins were identified by mass spectrometry and database queries: beta-crystallin B1, glyceraldehyde-3-phosphate dehydrogenase, carbonyl reductase (NADPH) 1, cDNA FLJ55253, gamma-crystallin D, GAS2-like protein 3, sorbitol dehydrogenase, DNA FLJ60282, phosphoglycerate kinase, and filensin.
Conclusion: The level of the ten proteins may play an important role in the development of liquefied after-cataracts.
Keywords: capsular block syndrome; liquefied after-cataract; liquid chromatography-tandem mass spectrometry.
Figures

Similar articles
-
Proteomic analysis of human age-related nuclear cataracts and normal lens nuclei.Invest Ophthalmol Vis Sci. 2011 Jun 13;52(7):4182-91. doi: 10.1167/iovs.10-7094. Invest Ophthalmol Vis Sci. 2011. PMID: 21436267
-
Quantitative proteomics analysis with iTRAQ in human lenses with nuclear cataracts of different axial lengths.Mol Vis. 2016 Jul 31;22:933-43. eCollection 2016. Mol Vis. 2016. PMID: 27559289 Free PMC article.
-
Proteomic analysis of water insoluble proteins from normal and cataractous human lenses.Mol Vis. 2007 Sep 14;13:1680-94. Mol Vis. 2007. PMID: 17893670
-
Multi-crystallin complexes exist in the water-soluble high molecular weight protein fractions of aging normal and cataractous human lenses.Exp Eye Res. 2008 Oct;87(4):356-66. doi: 10.1016/j.exer.2008.07.001. Epub 2008 Jul 10. Exp Eye Res. 2008. PMID: 18662688
-
Analysis of protein-protein interactions and proteomic profiles of normal human lenses.Curr Eye Res. 2010 Jul;35(7):605-19. doi: 10.3109/02713681003734833. Curr Eye Res. 2010. PMID: 20597647
References
-
- Dai Y, Huang Y, Xie L. Early postoperative capsular block syndrome analysis. Zhonghua Yan Shi Guang Xue Yu Shi Jue Ke Xue Za Zhi. 2008;10(3):225–227.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
