Thiopurine-induced mitotic catastrophe in Rad51d-deficient mammalian cells
- PMID: 28945288
- PMCID: PMC5771848
- DOI: 10.1002/em.22138
Thiopurine-induced mitotic catastrophe in Rad51d-deficient mammalian cells
Abstract
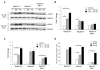
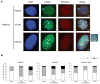
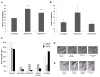
Thiopurines are part of a clinical regimen used for the treatment of autoimmune disorders and childhood acute lymphoblastic leukemia. However, despite these successes, there are also unintended consequences such as therapy-induced cancer in long-term survivors. Therefore, a better understanding of cellular responses to thiopurines will lead to improved and personalized treatment strategies. RAD51D is an important component of homologous recombination (HR), and our previous work established that mammalian cells defective for RAD51D are more sensitive to the thiopurine 6-thioguanine (6TG) and have dramatically increased numbers of multinucleated cells and chromosome instability. 6TG is capable of being incorporated into telomeres, and interestingly, RAD51D contributes to telomere maintenance, although the precise function of RAD51D at the telomeres remains unclear. We sought here to investigate: (1) the activity of RAD51D at telomeres, (2) the contribution of RAD51D to protect against 6TG-induced telomere damage, and (3) the fates of Rad51d-deficient cells following 6TG treatment. These results demonstrate that RAD51D is required for maintaining the telomeric 3' overhangs. As measured by γ-H2AX induction and foci formation, 6TG induced DNA damage in Rad51d-proficient and Rad51d-deficient cells. However, the extent of γ-H2AX telomere localization following 6TG treatment was higher in Rad51d-deficient cells than in Rad51d-proficient cells. Using live-cell imaging of 6TG-treated Rad51d-deficient cells, two predominant forms of mitotic catastrophe were found to contribute to the formation of multinucleated cells, failed division and restitution. Collectively, these findings provide a unique window into the role of the RAD51D HR protein during thiopurine induction of mitotic catastrophe. Environ. Mol. Mutagen. 59:38-48, 2018. © 2017 Wiley Periodicals, Inc.
Keywords: 6-thioguanine; H2AX; homologous recombination; mitotic catastrophe; telomeres.
© 2017 Wiley Periodicals, Inc.
Conflict of interest statement
All authors declare that there are no conflicts of interest.
Figures


References
-
- Armstrong MJ, Galloway SM. Mismatch repair provokes chromosome aberrations in hamster cells treated with methylating agents or 6-thioguanine, but not with ethylating agents. Mutation Research. 1997;373:167–178. - PubMed
-
- Broude EV, Loncarek J, Wada I, Cole K, Hanko C, Roninson IB, Swift M. Mitotic catastrophe in cancer therapy. In: Roninson IB, Brown JM, Bredesen DE, editors. Beyond apoptosis: Cellular outcomes of cancer therapy. CRC Press; 2008. pp. 307–320.
-
- Buermeyer AB, Wilson-Van Patten C, Baker SM, Liskay RM. The human mlh1 cdna complements DNA mismatch repair defects in mlh1-deficient mouse embryonic fibroblasts. Cancer research. 1999;59:538–541. - PubMed
-
- Castedo M, Perfettini JL, Roumier T, Andreau K, Medema R, Kroemer G. Cell death by mitotic catastrophe: A molecular definition. Oncogene. 2004;23:2825–2837. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

