Spontaneous, local diastolic subsarcolemmal calcium releases in single, isolated guinea-pig sinoatrial nodal cells
- PMID: 28945810
- PMCID: PMC5612473
- DOI: 10.1371/journal.pone.0185222
Spontaneous, local diastolic subsarcolemmal calcium releases in single, isolated guinea-pig sinoatrial nodal cells
Abstract
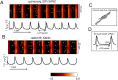
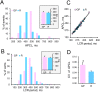
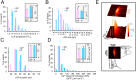
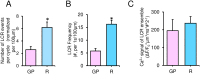
Uptake and release calcium from the sarcoplasmic reticulum (SR) (dubbed "calcium clock"), in the form of spontaneous, rhythmic, local diastolic calcium releases (LCRs), together with voltage-sensitive ion channels (membrane clock) form a coupled system that regulates the action potential (AP) firing rate. LCRs activate Sodium/Calcium exchanger (NCX) that accelerates diastolic depolarization and thus participating in regulation of the time at which the next AP will occur. Previous studies in rabbit SA node cells (SANC) demonstrated that the basal AP cycle length (APCL) is tightly coupled to the basal LCR period (time from the prior AP-induced Ca2+ transient to the diastolic LCR occurrence), and that this coupling is further modulated by autonomic receptor stimulation. Although spontaneous LCRs during diastolic depolarization have been reported in SANC of various species (rabbit, cat, mouse, toad), prior studies have failed to detect LCRs in spontaneously beating SANC of guinea-pig, a species that has been traditionally used in studies of cardiac pacemaker cell function. We performed a detailed investigation of whether guinea-pig SANC generate LCRs and whether they play a similar key role in regulation of the AP firing rate. We used two different approaches, 2D high-speed camera and classical line-scan confocal imaging. Positioning the scan-line beneath sarcolemma, parallel to the long axis of the cell, we found that rhythmically beating guinea-pig SANC do, indeed, generate spontaneous, diastolic LCRs beneath the surface membrane. The average key LCR characteristics measured in confocal images in guinea-pig SANC were comparable to rabbit SANC, both in the basal state and in the presence of β-adrenergic receptor stimulation. Moreover, the relationship between the LCR period and APCL was subtended by the same linear function. Thus, LCRs in guinea-pig SANC contribute to the diastolic depolarization and APCL regulation. Our findings indicate that coupled-clock system regulation of APCL is a general, species-independent, mechanism of pacemaker cell normal automaticity. Lack of LCRs in prior studies is likely explained by technical issues, as individual LCRs are small stochastic events occurring mainly near the cell border.
Conflict of interest statement
Figures
Similar articles
-
Heterogeneity of calcium clock functions in dormant, dysrhythmically and rhythmically firing single pacemaker cells isolated from SA node.Cell Calcium. 2018 Sep;74:168-179. doi: 10.1016/j.ceca.2018.07.002. Epub 2018 Jul 10. Cell Calcium. 2018. PMID: 30092494 Free PMC article.
-
High basal protein kinase A-dependent phosphorylation drives rhythmic internal Ca2+ store oscillations and spontaneous beating of cardiac pacemaker cells.Circ Res. 2006 Mar 3;98(4):505-14. doi: 10.1161/01.RES.0000204575.94040.d1. Epub 2006 Jan 19. Circ Res. 2006. PMID: 16424365
-
CaMKII-dependent phosphorylation regulates basal cardiac pacemaker function via modulation of local Ca2+ releases.Am J Physiol Heart Circ Physiol. 2016 Sep 1;311(3):H532-44. doi: 10.1152/ajpheart.00765.2015. Epub 2016 Jul 8. Am J Physiol Heart Circ Physiol. 2016. PMID: 27402669 Free PMC article.
-
Unique Ca2+-Cycling Protein Abundance and Regulation Sustains Local Ca2+ Releases and Spontaneous Firing of Rabbit Sinoatrial Node Cells.Int J Mol Sci. 2018 Jul 25;19(8):2173. doi: 10.3390/ijms19082173. Int J Mol Sci. 2018. PMID: 30044420 Free PMC article. Review.
-
Rhythmic Ca2+ oscillations drive sinoatrial nodal cell pacemaker function to make the heart tick.Ann N Y Acad Sci. 2005 Jun;1047:138-56. doi: 10.1196/annals.1341.013. Ann N Y Acad Sci. 2005. PMID: 16093492 Review.
Cited by
-
Dual Activation of Phosphodiesterase 3 and 4 Regulates Basal Cardiac Pacemaker Function and Beyond.Int J Mol Sci. 2021 Aug 5;22(16):8414. doi: 10.3390/ijms22168414. Int J Mol Sci. 2021. PMID: 34445119 Free PMC article. Review.
-
Self-Similar Synchronization of Calcium and Membrane Potential Transitions During Action Potential Cycles Predict Heart Rate Across Species.JACC Clin Electrophysiol. 2021 Nov;7(11):1331-1344. doi: 10.1016/j.jacep.2021.02.016. Epub 2021 Apr 28. JACC Clin Electrophysiol. 2021. PMID: 33933406 Free PMC article.
-
Intracellular Ca2+-Mediated Mechanisms for the Pacemaker Depolarization of the Mouse and Guinea Pig Sinus Node Tissue.Biomolecules. 2022 Feb 28;12(3):377. doi: 10.3390/biom12030377. Biomolecules. 2022. PMID: 35327569 Free PMC article.
-
Heart Rate, Hibernation, and the Power Law.JACC Clin Electrophysiol. 2021 Nov;7(11):1345-1347. doi: 10.1016/j.jacep.2021.04.006. JACC Clin Electrophysiol. 2021. PMID: 34794663 Free PMC article. No abstract available.
-
Dual Activation of Phosphodiesterases 3 and 4 Regulates Basal Spontaneous Beating Rate of Cardiac Pacemaker Cells: Role of Compartmentalization?Front Physiol. 2018 Oct 9;9:1301. doi: 10.3389/fphys.2018.01301. eCollection 2018. Front Physiol. 2018. PMID: 30356755 Free PMC article.
References
-
- Ju YK, Allen DG. The distribution of calcium in toad cardiac pacemaker cells during spontaneous firing. Pflugers Arch. 2000;441(2–3):219–27. - PubMed
-
- Rubenstein DS, Lipsius SL. Mechanisms of automaticity in subsidiary pacemakers from cat right atrium. Circulation research. 1989;64(4):648–57. - PubMed
-
- Rigg L, Terrar DA. Possible role of calcium release from the sarcoplasmic reticulum in pacemaking in guinea-pig sino-atrial node. Exp Physiol. 1996;81(5):877–80. - PubMed
-
- Allen DG, Blinks JR. Calcium transients in aequorin-injected frog cardiac muscle. Nature. 1978;273(5663):509–13. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

