Using the 4 Pillars™ Practice Transformation Program to increase adolescent human papillomavirus, meningococcal, tetanus-diphtheria-pertussis and influenza vaccination
- PMID: 28947346
- PMCID: PMC5647787
- DOI: 10.1016/j.vaccine.2017.09.039
Using the 4 Pillars™ Practice Transformation Program to increase adolescent human papillomavirus, meningococcal, tetanus-diphtheria-pertussis and influenza vaccination
Abstract
Objectives: To report the results of an intervention using the 4 Pillars™ Practice Transformation Program (4 Pillars™ Program) to increase adolescent vaccinations including human papillomavirus vaccine (HPV) and influenza vaccines, which remain underutilized in this population.
Study design: Eleven pediatric and family medicine practices, previously control sites from a randomized controlled cluster trial, with ≥50 adolescent patients participated. The 4 Pillars™ Program was the foundation of the intervention. De-identified demographic, office visit and vaccination data were derived from electronic medical record extractions for patients whose date of birth was 4/1/1997 to 4/1/2004 (ages 11-17years at baseline). Vaccination rates for HPV, influenza, tetanus-pertussis-diphtheria (Tdap) and meningococcal (MenACWY) vaccines were determined for all eligible patients pre- and post intervention (i.e., vaccination rates on 4/1/2015 and 4/30/2016).
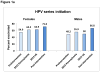
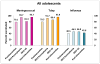
Results: Among 9473 patients ages 11-17years at baseline (4/1/2015), mean pre-intervention vaccination rates for HPV initiation and completion, meningococcal, Tdap and influenza vaccines were below national levels. Rates increased significantly post intervention (P<0.001) for HPV initiation which increased 17.1 percentage points (PP) from 51.4%; HPV completion increased 14.8PP from 30.7%, meningococcal vaccine uptake increased 16.6PP from 79.1%, Tdap vaccine uptake increased 14.6PP from 76.9%. Influenza vaccine uptake did not increase significantly (2.3PP from 40.1%). In the regression using generalized estimating equations, odds of vaccination were higher for younger, non-white adolescents for all vaccines; being in a smaller practice decreased the odds of Tdap vaccination but increased the odds of influenza vaccination.
Conclusion: Clinically and statistically significant improvements in HPV series initiation and completion, and meningococcal and Tdap vaccinations were observed in primary care practices implementing the 4 Pillars™ Practice Transformation Program. Clinical Trial Registry Number: NCT02165722.
Keywords: 4 Pillars; Adolescent vaccines; HPV vaccination; Tdap vaccination.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Figures

References
-
- US Department of Health and Human Services. [Accessed May 30, 2013];Healthy People 2020. 2012 http://www.healthypeople.gov/2020/topicsobjectives2020/objectiveslist.as....
-
- Perkins RB, Lin M, Silliman RA, Clark JA, Hanchate A. Why Are U.S. Girls Getting Meningococcal But Not Human Papilloma Virus Vaccines? Comparison of Factors Associated with Human Papilloma Virus and Meningococcal Vaccination Among Adolescent Girls 2008 to 2012. Women's Health Issues. 2015;25(2):97–104. - PubMed
-
- Jeudin P, Liveright E, Del Carmen MG, Perkins RB. Race, ethnicity, and income factors impacting human papillomavirus vaccination rates. Clinical therapeutics. 2014;36(1):24–37. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

