Aging-Impaired Filamentous Actin Polymerization Signaling Reduces Alveolar Macrophage Phagocytosis of Bacteria
- PMID: 28947541
- PMCID: PMC5679440
- DOI: 10.4049/jimmunol.1700140
Aging-Impaired Filamentous Actin Polymerization Signaling Reduces Alveolar Macrophage Phagocytosis of Bacteria
Abstract
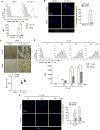
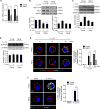
In elderly patients, bacterial infection often causes severe complications and sepsis. Compared to younger patients, older patients are more susceptible to sepsis caused by respiratory infection. Macrophage (Mϕ) phagocytosis of bacteria plays a critical role in the clearance of pathogens and the initiation of immune responses. It has been suggested that Mϕ exhibit age-related functional alterations, including reduced chemotaxis, phagocytosis, antibacterial defense, and the ability to generate reactive oxygen species. However, the mechanisms behind these changes remain unclear. The present study sought to determine changes in bacterial phagocytosis in aging alveolar Mϕ (AMϕ) and the underlying mechanisms. We show that bacteria initiate cytoskeleton remodeling in AMϕ through interaction with macrophage receptor with collagenous structure (MARCO), a bacterial scavenger receptor. This remodeling, in turn, promotes enhanced cell surface expression of MARCO and bacterial phagocytosis. We further demonstrate that Rac1-GTP mediates MARCO signaling and activates actin-related protein-2/3 complex, an F-actin nucleator, thereby inducing F-actin polymerization, filopodia formation, and increased cell surface expression of MARCO, all of which are essential for the execution of bacteria phagocytosis. However, AMϕ isolated from aging mice exhibit suppressed Rac1 mRNA and protein expression, which resulted in decreases in Rac1-GTP levels and actin-related protein-2/3 activation, as well as subsequent attenuation of F-actin polymerization, filopodia formation, and cell surface expression of MARCO. As a result, bacterial phagocytosis in aging AMϕ is decreased. This study highlights a previously unidentified mechanism by which aging impairs Mϕ phagocytosis of bacteria. Targeting these pathways may improve outcomes of bacterial infection in elderly patients.
Copyright © 2017 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures



References
-
- Kline KA, Bowdish DM. Infection in an aging population. Curr Opin Microbiol. 2016;29:63–67. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

