Bio-Inspired Carbon Monoxide Sensors with Voltage-Activated Sensitivity
- PMID: 28952172
- PMCID: PMC5658252
- DOI: 10.1002/anie.201707491
Bio-Inspired Carbon Monoxide Sensors with Voltage-Activated Sensitivity
Abstract
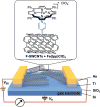
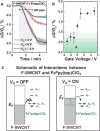
Carbon monoxide (CO) outcompetes oxygen when binding to the iron center of hemeproteins, leading to a reduction in blood oxygen level and acute poisoning. Harvesting the strong specific interaction between CO and the iron porphyrin provides a highly selective and customizable sensor. We report the development of chemiresistive sensors with voltage-activated sensitivity for the detection of CO comprising iron porphyrin and functionalized single-walled carbon nanotubes (F-SWCNTs). Modulation of the gate voltage offers a predicted extra dimension for sensing. Specifically, the sensors show a significant increase in sensitivity toward CO when negative gate voltage is applied. The dosimetric sensors are selective to ppm levels of CO and functional in air. UV/Vis spectroscopy, differential pulse voltammetry, and density functional theory reveal that the in situ reduction of FeIII to FeII enhances the interaction between the F-SWCNTs and CO. Our results illustrate a new mode of sensors wherein redox active recognition units are voltage-activated to give enhanced and highly specific responses.
Keywords: Carbon monoxide; carbon nanotubes; iron porphyrin; sensors; voltage-activated.
© 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
The authors declare the following competing financial interest(s): A patent has been filed on this technology.
Figures




References
-
- Raub JA, Mathieu-Nolf M, Hampson NB, Thom SR. Toxicology. 2000;145:1–14. - PubMed
-
- Occupational Safety and Health Administration. Carbon Monoxide In Workplace Atmospheres (Direct-Reading Monitor) 1993.
-
- Gonzales FJ. Pharmacol. Rev. 1985;40:243–288. - PubMed
-
- Klingenberg M. Arch. Biochem. Biophys. 1958;75:376–386. - PubMed
-
- Omura T, Sato R. J. Biol. Chem. 1964;239:2370–2378. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

