Combined replacement effects of human modified β-hexosaminidase B and GM2 activator protein on GM2 gangliosidoses fibroblasts
- PMID: 28955902
- PMCID: PMC5613250
- DOI: 10.1016/j.bbrep.2016.04.012
Combined replacement effects of human modified β-hexosaminidase B and GM2 activator protein on GM2 gangliosidoses fibroblasts
Abstract
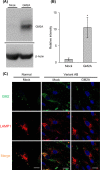
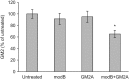
GM2 gangliosidoses are autosomal recessive lysosomal storage diseases (LSDs) caused by mutations in the HEXA, HEXB and GM2A genes, which encode the human lysosomal β-hexosaminidase (Hex) α- and β-subunits, and GM2 activator protein (GM2A), respectively. These diseases are associated with excessive accumulation of GM2 ganglioside (GM2) in the brains of patients with neurological symptoms. Here we established a CHO cell line overexpressing human GM2A, and purified GM2A from the conditioned medium, which was taken up by fibroblasts derived from a patient with GM2A deficiency, and had the therapeutic effects of reducing the GM2 accumulated in fibroblasts when added to the culture medium. We also demonstrated for the first time that recombinant GM2A could enhance the replacement effect of human modified HexB (modB) with GM2-degrading activity, which is composed of homodimeric altered β-subunits containing a partial amino acid sequence of the α-subunit, including the GSEP loop necessary for binding to GM2A, on reduction of the GM2 accumulated in fibroblasts derived from a patient with Tay-Sachs disease, a HexA (αβ heterodimer) deficiency, caused by HEXA mutations. We predicted the same manner of binding of GM2A to the GSEP loop located in the modified HexB β-subunit to that in the native HexA α-subunit on the basis of the x-ray crystal structures. These findings suggest the effectiveness of combinational replacement therapy involving the human modified HexB and GM2A for GM2 gangliosidoses.
Keywords: CI-M6PR, cation-independent M6P receptor; CM, conditioned medium; ERT, enzyme replacement therapy; Enzyme replacement therapy; GM2, GM2 ganglioside; GM2A, GM2 activator protein; Gm2 activator protein; Gm2 gangliosidosis; Hex, β-hexosaminidase; LAMP-1, lysosomal associated membrane protein 1; LSD, lysosomal storage disease; Lysosomal storage disease; M6P, mannose-6-phosphate; SD, Sandhoff disease; TSD, Tay-Sachs disease; modB, modified HexB; β-hexosaminidase.
Figures


References
-
- Gravel R.A., Kaback M.M., Proia R.L. The GM2 gangliosidoses. In: Scriver C.R., Beaudet A.L., Sly W.S., Valle D., editors. The Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill; New York: 2001. pp. 3827–3876.
-
- Wu Y.Y., Lockyer J.M., Sugiyama E. Expression and specificity of human GM2 activator protein. J. Biol. Chem. 1994;269:16276–16283. - PubMed
-
- Smiljanic-Georgijev N., Rigat B., Xie B. Characterization of the affinity of the GM2 activator protein for glycolipids by a fluorescence dequenching assay. Biochim. Biophys. Acta. 1997;1339:192–202. - PubMed
-
- Barton N.W., Brady R.O., Dambrosia J.M. Replacement therapy for inherited enzyme deficiency–macrophage-targeted glucocerebrosidase for Gaucher's disease. N. Engl. J. Med. 1991;324:1464–1470. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

