Proteolytic processing and inactivation of CCL2/MCP-1 by meprins
- PMID: 28955950
- PMCID: PMC5613766
- DOI: 10.1016/j.bbrep.2016.08.019
Proteolytic processing and inactivation of CCL2/MCP-1 by meprins
Abstract
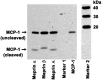
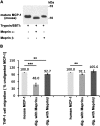
Monocyte chemotactic protein 1 (CCL2/MCP-1) is a small chemokine involved in the recruitment and trafficking of mononuclear immune cells to inflammation sites. Our studies demonstrate that the metalloendopeptidases meprin A (purified from kidney cortex), recombinant meprin α, and recombinant meprin β can all process CCL2/MCP-1. The cleavage sites were determined by amino acid sequencing and mass spectrometry analysis of the generated products, and the biological activity of the products was evaluated by chemotactic migration assay using THP-1 cells. The cleavage sites generated by the meprin isoforms revealed that meprin A and meprin α cleaved the N-terminal domain of mouse CCL2/MCP-1 at the Asn6 and Ala7 bond, resulting in significant reduction in the chemotactic activity of the cleaved CCL2/MCP-1. Meprin β was unable to cleave the N-terminus of mouse CCL2/MCP-1 but cleaved the C-terminal region between Ser74 and Glu75. Human CCL2/MCP-1 that lacks the murine C-terminal region was also cleaved by meprin α at the N-terminus resulting in significant loss of CCL2/MCP-1 biological activity, whereas meprin β did not affect the biological activity. These studies suggest that meprin α and meprin β may play important roles in regulating the CCL2/MCP-1 chemokine activity during inflammation.
Keywords: Chemokine; Inflammation; Meprin A; Meprin α; Meprin β; Metalloproteinase; Monocyte chemotactic protein 1.
Figures


Similar articles
-
Role of meprin metalloproteinases in cytokine processing and inflammation.Cytokine. 2019 Feb;114:18-25. doi: 10.1016/j.cyto.2018.11.032. Epub 2018 Dec 20. Cytokine. 2019. PMID: 30580156 Free PMC article. Review.
-
Isoform-specific interactions between meprin metalloproteases and the catalytic subunit of protein kinase A: significance in acute and chronic kidney injury.Am J Physiol Renal Physiol. 2015 Jan 1;308(1):F56-68. doi: 10.1152/ajprenal.00167.2014. Epub 2014 Oct 29. Am J Physiol Renal Physiol. 2015. PMID: 25354939 Free PMC article.
-
Maturation of secreted meprin alpha during biosynthesis: role of the furin site and identification of the COOH-terminal amino acids of the mouse kidney metalloprotease subunit.Arch Biochem Biophys. 1998 Jan 1;349(1):192-200. doi: 10.1006/abbi.1997.0453. Arch Biochem Biophys. 1998. PMID: 9439598
-
Villin and actin in the mouse kidney brush-border membrane bind to and are degraded by meprins, an interaction that contributes to injury in ischemia-reperfusion.Am J Physiol Renal Physiol. 2011 Oct;301(4):F871-82. doi: 10.1152/ajprenal.00703.2010. Epub 2011 Jul 27. Am J Physiol Renal Physiol. 2011. PMID: 21795642 Free PMC article.
-
The metalloproteases meprin α and meprin β: unique enzymes in inflammation, neurodegeneration, cancer and fibrosis.Biochem J. 2013 Mar 1;450(2):253-64. doi: 10.1042/BJ20121751. Biochem J. 2013. PMID: 23410038 Free PMC article. Review.
Cited by
-
Role of meprin metalloproteinases in cytokine processing and inflammation.Cytokine. 2019 Feb;114:18-25. doi: 10.1016/j.cyto.2018.11.032. Epub 2018 Dec 20. Cytokine. 2019. PMID: 30580156 Free PMC article. Review.
-
Meprin β metalloproteases associated with differential metabolite profiles in the plasma and urine of mice with type 1 diabetes and diabetic nephropathy.BMC Nephrol. 2019 Apr 25;20(1):141. doi: 10.1186/s12882-019-1313-2. BMC Nephrol. 2019. PMID: 31023251 Free PMC article.
-
Undiagnosed Kidney Injury in Uninsured and Underinsured Diabetic African American Men and Putative Role of Meprin Metalloproteases in Diabetic Nephropathy.Int J Nephrol. 2018 Apr 29;2018:6753489. doi: 10.1155/2018/6753489. eCollection 2018. Int J Nephrol. 2018. PMID: 29854459 Free PMC article.
-
Helical ultrastructure of the metalloprotease meprin α in complex with a small molecule inhibitor.Nat Commun. 2022 Oct 19;13(1):6178. doi: 10.1038/s41467-022-33893-7. Nat Commun. 2022. PMID: 36261433 Free PMC article.
-
Inhibition of miR-155-5p Exerts Anti-Fibrotic Effects in Silicotic Mice by Regulating Meprin α.Mol Ther Nucleic Acids. 2020 Mar 6;19:350-360. doi: 10.1016/j.omtn.2019.11.018. Epub 2019 Nov 26. Mol Ther Nucleic Acids. 2020. PMID: 31877411 Free PMC article.
References
-
- Adams D.H., Lloyd A.R. Chemokines: leucocyte recruitment and activation cytokines. Lancet. 1997;349:490–495. - PubMed
-
- Moser B., Loetscher P. Lymphocyte traffic control by chemokines. Nat. Immunol. 2001;2:123–128. - PubMed
-
- Hsieh K.H., Chou C.C., Chiang B.L. Immunotherapy suppresses the production of monocyte chemotactic and activating factor and augments the production of IL-8 in children with asthma. J. Allergy Clin. Immunol. 1996;98:580–587. - PubMed
-
- Chizzolini C., Brembilla N.C., Montanari E., Truchetet M.E. Fibrosis and immune dysregulation in systemic sclerosis. Autoimmun. Rev. 2011;10:276–281. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

